Introduction
Tannerella forsythia, Treponema denticola, Prevotella intermedia, Fusobacterium nucleatum, and Capnocytophaga species are major periodontal pathogens. These bacteria have been found in increased numbers in areas with active periodontal destruction. There are many other microorganisms also which are actively involved in periodontal disease progression but have not been well investigated. In the present discussion, we shall focus on the above-said bacteria and their disease-causing capability, specifically during periodontal tissue breakdown in periodontitis.
Tannerella forsythia
This microorganism is an anaerobic, fusiform bacterium that was first isolated in the mid-1970s at The Forsyth Institute from subjects with progressing advanced periodontitis and was described as ‘Fusiform bacteroides’ by Tanner et al. (1979) 212. It belongs to the Cytophaga-Bacteroides family and was initially named as Bacteroides forsythus. Later on, it was reclassified as Tannerella forsythia by Sakamoto et al. (2002) 213, based on 16S rRNA phylogenetic analysis. The major problem with the isolation and cultivation of this microorganism is that it has anaerobic growth requirements for cultural detection.
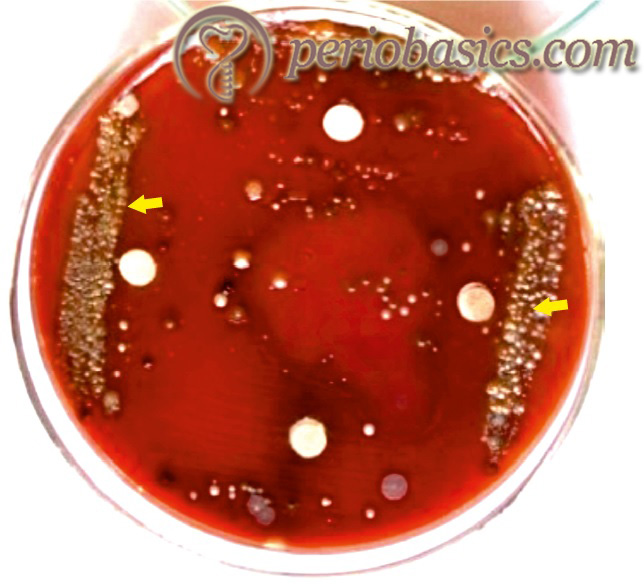
Most of the studies done on this microorganism are based on non-cultural approaches (immunoassays and DNA-based assays). There are many studies which have implicated Tannerella forsythia as a causative agent for progressive clinical attachment loss associated with periodontitis 6, 7, 40, 214-215. So far, the virulence factors identified for T. forsythia include,
- Trypsin-like proteases 216
- PrtH proteases 217,
- Sialidases SiaH 218, 219 and NanH 220,
- A leucine-rich repeat cell-surface-associated and secreted protein BspA 221,
- An apoptosis-inducing activity 222,
- α-D-glucosidase and N-acetyl-beta-glucosaminidase 223,
- A hemagglutinin 224,
- Components of the bacterial S-layer 225, 226, and
- Methylglyoxal production 227.
Following is a brief description of a few of these virulence factors:
Proteases
T. forsythia is an asaccharolytic microorganism and requires the presence of peptides and free amino acids for its growth. It procures these essential substrates from the host by the degradation of host proteins. Two proteases produced by T. forsythia have been identified which perform this function. These are a trypsin-like protease and PrtH proteases. Along with the degradation of host proteins for their growth, these also act as important virulence factors for host invasion in multiple ways which include 228,
- By causing periodontal connective tissue destruction.
- By activating host degradative enzymes.
- By cleaving components involved in innate (cytokines/ chemokines, complement factors) immunity.
- By modifying host cell proteins to expose cryptotopes for bacterial colonization.
Surface-layer associated glycoproteins
The surface components of T. forsythia play a vital role in its virulence. These include,
S-layer:
The microorganism possesses a surface layer which is made up of serrated structural subunits (about 10 nm wide and 10 nm high) in either oblique or tetragonal lattices 229. This layer is usually described as S-layer. It consists of two high molecular weight glycoproteins (220 and 210 kDa). These glycoproteins are encoded by tfsA and tfsB genes, respectively 230-231. Along with providing protective shielding, this layer also helps in …….Contents available in the book…….Contents available in the book…….Contents available in the book…….Contents available in the book…….Contents available in the book…..
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Leucine-rich repeat BspA protein:
This is a surface as well as secreted protein that act as an important virulence factor for T. forsythia. Short term used for Bacteroides surface protein A is BspA. This protein has the ability to interact with host factors and/or components. It has been shown to bind to the extracellular matrix component, fibronectin and the clotting factor fibrinogen 221. BspA also facilitates bacterial adherence and invasion into epithelial cells 233. Furthermore, this protein has also been shown to mediate surface interaction with F. nucleatum during biofilm formation 234. By activating the TLR-2-dependent pathway, BspA has been shown to trigger the release of bone-resorbing pro-inflammatory cytokines from monocytes 235 and IL-8 from gingival epithelial cells 236. Hence, it can be concluded that BspA due to its interaction with host immune response plays an important role in the pathogenesis of periodontal tissue breakdown.
Surface lipoproteins
The surface lipoproteins are involved in the release of pro-inflammatory cytokines (IL-6 and TNF-α) from human gingival fibroblasts and monocytic cells 237. Furthermore, these also have been shown to induce cellular apoptosis 237. The cell apoptosis induced by surface lipoproteins is triggered by activation of caspase-8 which is an initiator of the caspase cascade in apoptosis.
Glycosidic activity
T. forsythia expresses a variety of glycosidases including neuraminidases SiaHI 219, neuraminidases NanH 220, α-D-glucosidase and N-acetyl β-D glucosaminidase (hexA) 223. These enzymes are responsible for the degradation of various host oligosaccharides and proteoglycans, the products of which provides nutrition to various bacterial species in the plaque bacterial community. Furthermore, the exposure of glycosidases also results in the exposure of epitopes for adherence of bacteria. This glycosidic activity also yields to the accumulation of a toxic methylglyoxal product in vitro, which is theorized to contribute to tissue damage in hosts affected by periodontitis and colonized by this pathogen.
Treponema denticola
Oral Treponemes are the members of the Spirochaetes phylum having three families (Spirochaetaceae, Brachyspiraceae, and Leptospiraceae) and nine genera. Out of these, genus Treponema includes the causative agents of syphilis and periodontal pathogens. Spirochetes have a distinct shape and structure. Their length varies from 5 to 20 µm and diameter ranges from 0.1 to 0.5 µm. Structurally, they are similar to Gram-positive bacteria consisting of inner and outer membranes. However, most of the species lack lipopolysaccharide (LPS) and instead have lipoproteins and glycolipids 238, 239. The ultrastructure of spirochetes is characterized by the presence of internal flagella known as endoflagella. Two distinct bundles are formed by endoflagella within the periplasmic space which insert into the inner membrane at opposite ends of the cell. These bundles extend up to two-third the length of the cell, wrapping around the inner membrane in a right-handed manner.

Among all the oral spirochetes, one of the most studied is T. denticola which possesses the features needed for adherence, invasion, and damage of the periodontal tissues. This periodontal pathogen mostly inhabits the deeper periodontal pockets and is not an early colonizer of subgingival plaque 240. It is typically found in dense subgingival bacterial biofilms at the interface of the biofilms and the gingival epithelium 241, 242. It specifically binds to both …….Contents available in the book…….Contents available in the book…….Contents available in the book…….Contents available in the book…….Contents available in the book…… These include,
- Major sheath protein (Msp).
- Leucine-rich repeat proteins.
- Transposases.
- Lipooligosaccharides (LOS).
- Periplasmic flagella.
- Dentilisin.
- Cystalysin.
- Trypsin-like proteases.
- Lipoproteins.
- Outer sheath vesicles.
- Factor H binding protein.
- Immunomodulation factors.
These virulence factors are discussed as follows,
Major sheath protein (Msp)
The major outer sheath protein (Msp) is a primary virulence determinant in T. denticola. It is the principal immunogenic surface antigen that forms oligomeric protein complexes in the outer membrane of this bacterium 246. Msp has been shown to have matrix protein binding properties in vitro 247 and can cause transient transmembrane ion fluxes and depolarization of host cells 248, 249. Hence, it has been implicated as a mediator of the interaction between the spirochetes and the gingival epithelium in periodontal diseases.
Leucine-rich repeat proteins
These proteins are members of the cytolethal distending toxin (Cdt) family of proteins that are secreted and attached to the cell surface. These are protein-interaction motifs of 20-29 residues consisting of a high proportion of leucine residues. These are primarily involved in the formation of a structural framework for protein-protein interactions. The leucine-rich repeat protein (LrrA) plays an important role in the binding of T. denticola to T. forsythia 250. Gene lrrA, encoding a leucine-rich repeat protein, LrrA, that contains eight consensus tandem repeats of 23 amino acid residues, has been identified in T. denticola ATCC 35405 250. Furthermore, it has also been shown that leucine-rich repeat protein LrrA plays a role in the attachment and penetration of human epithelial cells 250.
Transposases
Transposases are a family of enzymes that “cut and paste” mobile genetic elements from one position to another within the genome. It has been observed that 25 out of 35 putative transposases genes were upregulated in a biofilm as compared to the planktonic cell, indicating that these participate in novel gene-regulatory mechanisms in a biofilm 251.
Lipooligosaccharides (LOS)
The outer sheath of T. denticola does not have a typical lipopolysaccharide framework. Instead, it contains lipooligosaccharide (LOS) which consist of two regions Lipid A and R polysaccharide (core glycolipid) and lacks the O antigen. LOS has been shown to stimulate the activation of NO (nitric oxide) and TNF-α in LPS-responsive and -non-responsive mouse macrophages. The macrophage activation by LOS is similar to lipopolysaccharide activation 252.
Periplasmic flagella
T. denticola has usually two periplasmic flagellae attached at each end. The flagellar filament of T. denticola consists of 3 core proteins: FlaB1, FlaB2 and FlaB3. These flagella facilitate translocation of the bacteria in highly viscous environments. Furthermore, it has been shown that periplasmic flagella do not get immobilized by flagella-specific antibodies produced by the host response generated against infection 253.
Dentilisin
It is a protease located on the cell surface of T. denticola that cleaves at phenylalanyl/alanyl and prolyl/alanyl bonds 254. The active site of these serine proteases contains a His-Asp-Ser motif in chymotrypsin that is believed to be responsible for catalyzing the hydrolysis of peptide bonds. It contributes to …….Contents available in the book…….Contents available in the book…….Contents available in the book…….Contents available in the book…….Contents available in the book…..
Cystalysin
It is a hemolytic protein capable of oxidizing hemoglobin and causing lysis of human erythrocytes 255. The production of H2S is considered to be the major function of cystalysin, which can thus be regarded as an important virulence factor 256, 257. It contributes to oral malodor.
Trypsin-like proteases
The enzyme responsible for trypsin-like activity in T. denticola is oligopeptidase B which is encoded by opdB (TDE2140). This enzyme has been shown to cleave C-terminal to Arg-residues 258. The exact role of this enzyme in the virulence of T. denticola still remains to be clarified.
Lipoproteins
These are the most abundant membrane-associated proteins found in spirochetes. Twenty lipoproteins have been detected in T. denticola in a study by Veith et al. (2009) 259 including 2 hemin-binding proteins (HbpA, HbpB), 2 putative extra-cellular solute-binding lipoproteins, 4 putative oligopeptide /dipeptide ABC transporter peptide-binding proteins, the dentilisin complex-associated polypeptide (PrcA) and 11 uncharacterized putative lipoproteins. The exact role of these lipoproteins in the virulence of T. denticola still needs to be clarified.
Outer sheath vesicles
The outer sheath vesicles are produced by bacteria in response to changes in the external environment 260, 261. These vesicles are potent virulence factors because they possess adhesins, toxins, and proteolytic enzymes, which facilitate bacterial invasion and aggregation, are cytotoxic, and can modulate the host immune response 262. Furthermore, these vesicles help in bacterial aggregation by delivering proteases and toxins which are active against other bacterial species, thus facilitating to secure a niche in the competitive environment of subgingival plaque 263, 264. These vesicles also facilitate remote delivery of labile signaling molecules and prevent their degradation by other microorganisms 265. The outer sheath vesicles of T. denticola have also been implicated in tissue invasion 262, 266. These have been shown to disrupt the tight junctions in epithelial cell monolayers, hence facilitating connective tissue invasion 267.
Factor H binding protein
T. denticola possesses a small surface-exposed lipoprotein, FhbB. This lipoprotein binds complement regulatory proteins of the factor H (FH) family. It cleaves factor H into sub-fragment of ~50 kDa. This lipoprotein, also down-regulates the production of C3b. Thus, this factor is involved in the immunomodulation.
Immunomodulation factors
T. denticola can evade various aspects of innate immunity by preventing the efficient binding of antimicrobial peptides, such as β-defensins. β-defensins are antibacterial peptides which play an important role in the initial host defense against microbial attack. These small cationic peptides interact with negatively charged cell wall components of bacteria and fungi disrupting membrane integrity.
It has also been demonstrated that young patients with generalized severe periodontitis exhibited lower serum levels of antibodies against T. denticola as compared to those in healthy subjects 268. The diminished antibody response against T. denticola in patients with aggressive periodontitis is suggestive of its immunomodulatory properties. Furthermore, T. denticola has also been reported to affect the migration of neutrophils 270.
Prevotella intermedia
P. intermedia are Gram-negative, black-pigmented, obligate anaerobic rods which have been implicated as putative periodontal pathogens 51, 52, 281-284. These are called as “black pigmented bacteria” because they form shiny and smooth colonies with gray, light brown or black color on blood agar plate 285. P. intermedia were formerly known as Bacteroides intermedia or Bacteroides melaninogenicus. They co-aggregate with other Gram-negative oral bacteria such as P. gingivalis and Actinomyces viscosus during plaque formation. This bacterial species appears to have multiple virulence factors exhibited by P. gingivalis and has been shown to induce mixed infections on injection in laboratory animals 286.
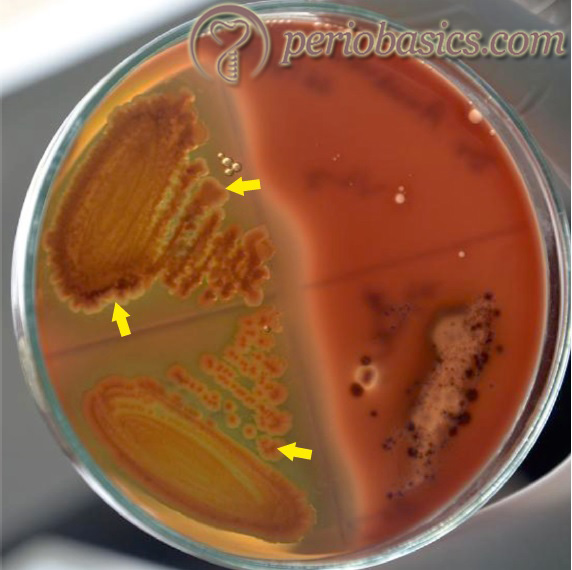
Furthermore, it has also been shown to invade oral epithelial cells in vitro 279. Along with periodontal infections, P. intermedia has also been reported to colonize in the respiratory tract and has been found to be associated with cystic fibrosis and chronic bronchitis 287, 288. It utilizes estradiol and progesterone as a growth factor, instead of vitamin K 289. There are various virulence factors associated with P. intermedia. Some of these are,
- Lipopolysaccharides
- Proteinases
- Immunomodulators
Lipopolysaccharides
LPS is a major constituent of the outer membrane of P. intermedia. It is an important virulence factor which can trigger a number of host cells, especially mononuclear phagocytes, to produce and release a wide variety of pharmacologically active mediators, including IL-1β, IL-6, IL-8, and, most importantly, tumor necrosis factor-α (TNF-α) 290, IL-10 291, IL-6 and IL-8 292, 293.
Proteinases
Proteinases produced by P. intermedia contribute in tissue destruction and increased availability of nutrients. The proteinases produced by P. intermedia display trypsin-like and dipeptidyl peptidase activities 294. These proteinases have also been shown to possess properties of cysteine proteinases 295-297.
Immunomodulators
Interpain A is a secreted protein composed of 868 amino acid residues 298. It has been shown to degrade C3 complement of complement system 299. Hence, P. intermedia can modulate the innate immune response which facilitates its survival in the host tissues.
Fusobacterium nucleatum
Fusobacterium nucleatum is an anaerobic oral commensal and a periodontal pathogen. It is a Gram-negative bacterium that does not create spores and is non-motile. It belongs to the family Bacteroidaceae. Other members of this family found in the oral cavity include Fusobacterium necrophorum, and Fusobacterium varium. Fusobacteria are spindle-shaped, but may also appear polymorphous and can take on many other morphologies, including irregular spherical swollen cells and linear cells. These are obligate anaerobes and can be grown in aerobic conditions on agar plates when 5-10% CO2 is added to the culture conditions. Fusobacteria require rich media for growth and usually grow well in media containing trypticase, peptone, or yeast extract.
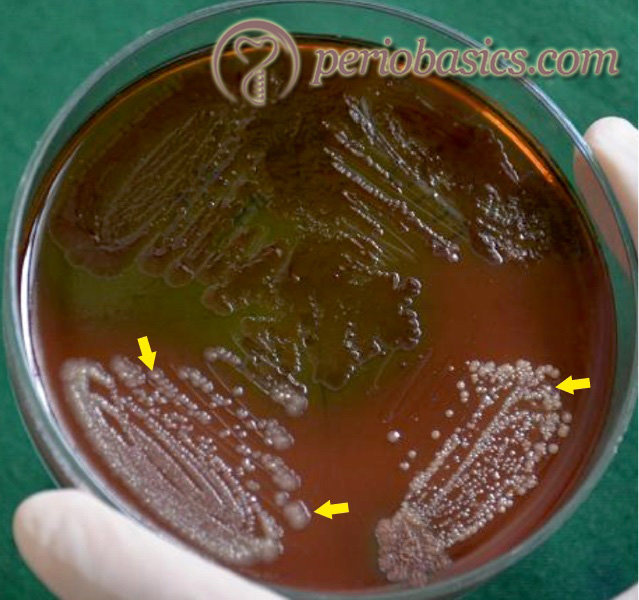
Most of the strains of F. nucleatum produce H2S and can agglutinate red blood cells. The ability of the bacteria to lyse erythrocytes and release hemoglobin is considered a virulence determinant since it provides an iron source to F. nucleatum and other periodontopathogens. The outer membrane of F. nucleatum contains a number of adhesins that are involved in adherence to salivary proteins, bacteria, and host cells. The bacterium is invasive and …….Contents available in the book…….Contents available in the book…….Contents available in the book…….Contents available in the book…….Contents available in the book…..
F. nucleatum is a prominent component of plaque biofilm quantitatively and is one of the first Gram-negative species to become established in plaque. The bacteria play a key role in the subgingival biofilm formation by bridging the early colonizers (Streptococci and Actinomyces) and the late colonizers (Porphyromonas gingivalis, Tannerella forsythia, Treponema denticola). Thus, F. nucleatum can be used as a key target for controlling biofilm formation.
Capnocytophaga species
Capnocytophaga species are long, thin Gram-negative bacilli that are slow-growing, with gliding ability on agar media. They are facultatively anaerobic and require enrichment with CO2 (5-10%) for optimum growth (capnophilic). The organisms grow best at 35 to 37 °C on either blood or chocolate agar. Microscopic observation demonstrates a high degree of polymorphism, with a variation in the size and appearance of the bacteria depending on the strain and culture conditions. Many strains produce a yellow-brown pigment. The organisms are relatively fastidious and slowly growing, so that colonies become apparent only after prolonged incubation (48 to 72 hours). Figure 5.10 demonstrates Capnocytophaga colonies on blood agar.
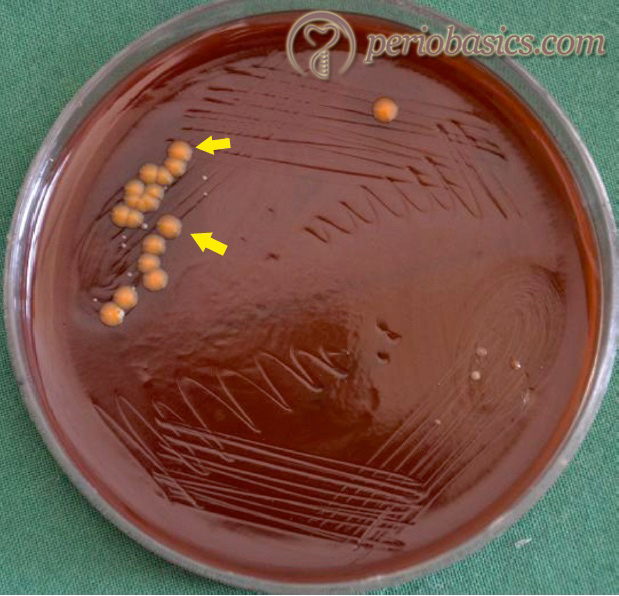
Capnocytophaga species are among the normal flora of the oral cavity and are potential etiological agents in periodontitis with rapid rate of progression. The common Capnocytophaga species isolated from the human oral cavity include C. gingivalis, C. granulosa, C. hemolytica, C. leadbetteri, C. ochracea, and C. sputigena. The pathogenic potential of Capnocytophaga species have been attributed in part to the production of tissue-destroying hydrolytic enzymes. The aminopeptidase of Capnocytophaga is an important virulence factor of the bacteria. In addition to aminopeptidase activity, a trypsin-like protease has been identified, purified and characterized. Capnocytophaga species are often associated with …….Contents available in the book…….Contents available in the book…….Contents available in the book…….Contents available in the book…….Contents available in the book…..
Conclusion
In the above discussion, we studied about some well-investigated bacteria that have been associated with periodontal tissue breakdown. It must be understood here that there are many micro-organisms that have been associated with periodontal disease progression. Only few out of them have been studied extensively. It is a field in periodontics that is wide open for research. Hence, in the future, many other micro-organisms are expected to be investigated for their role in periodontal tissue breakdown.
References
References available in the hard copy of the website.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

