Introduction to periodontal disease progression
There has been a lot of research to understand the exact nature of periodontal diseases. Various researchers have tried to understand the rate of diseases progression in various forms of periodontitis. In the past, several patterns of disease progression have been described 135-138. The patterns of tissue destruction in the progression of periodontal diseases have been explained with the help of various models including,
- Continuous model: This model explained that the periodontal disease progression occurs in a slow, steady and progressive manner. However, this model did not explain the rapid progression and periodic remission of the disease activity.
- Episodic burst model: Subsequent studies explained the irregular periods of exacerbation and remission. They explained that disease progression occurs as an episodic burst of activity with periods of remission.
- Synchronous burst model: It explained that the periodontal diseases progress with periods of exacerbation and remission during a defined period.
- Epidemiologic model: This model proposed that the disease progression is continuous with aging and it depends only on the duration of the process.
- Brownian motion or stochastic model: According to this model, random periods of sharp bursts and/or remission can occur, but the underlying disease activity remains constant.
- Random walking model: When observed at regular intervals this model is similar to the Brownian motion model.
- Fractal model: It is a multi-factorial model simulating disease advancing with age with burst and remission.
Although, many models have been proposed, but it has been recognized that linear disease progression can occur and there may be episodes of tissue destruction 139, 140.
Recent models of periodontal disease progression
It is a well-established fact that periodontal diseases are multi-factorial diseases with microbial etiology being their primary etiology. In the past various models of periodontal disease progression have been described (discussed above). The newer models of periodontal disease progression came into the picture with the introduction of the “non-linear model” of periodontal disease progression by Kenneth S. Kornman in 1997 27.
Non-linear model by Kornman
According to this model (Korman 1997) 27, host immuno-inflammatory mechanisms are activated by bacterial products. As a result, the periodontal disease progression depends on the interaction between the host and the microorganisms. Furthermore, the clinical expressions of disease are primarily determined by genetic and environmental factors that modify the host response. Each combination of genetic variations and environmental factors may define a specific gene expression pattern. This model explains various factors responsible for periodontal disease progression; however, some of the concepts in this model have become obsolete in the recent scenario leading to the development of advanced models for progression of periodontal disease.
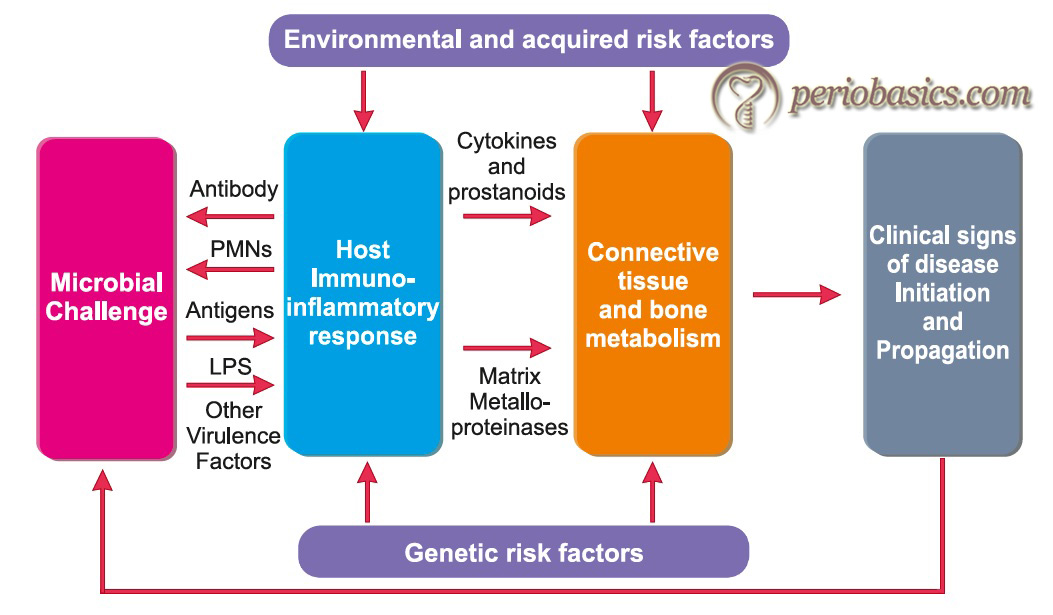
Multilevel hierarchical and biologic systems model by Kornman:
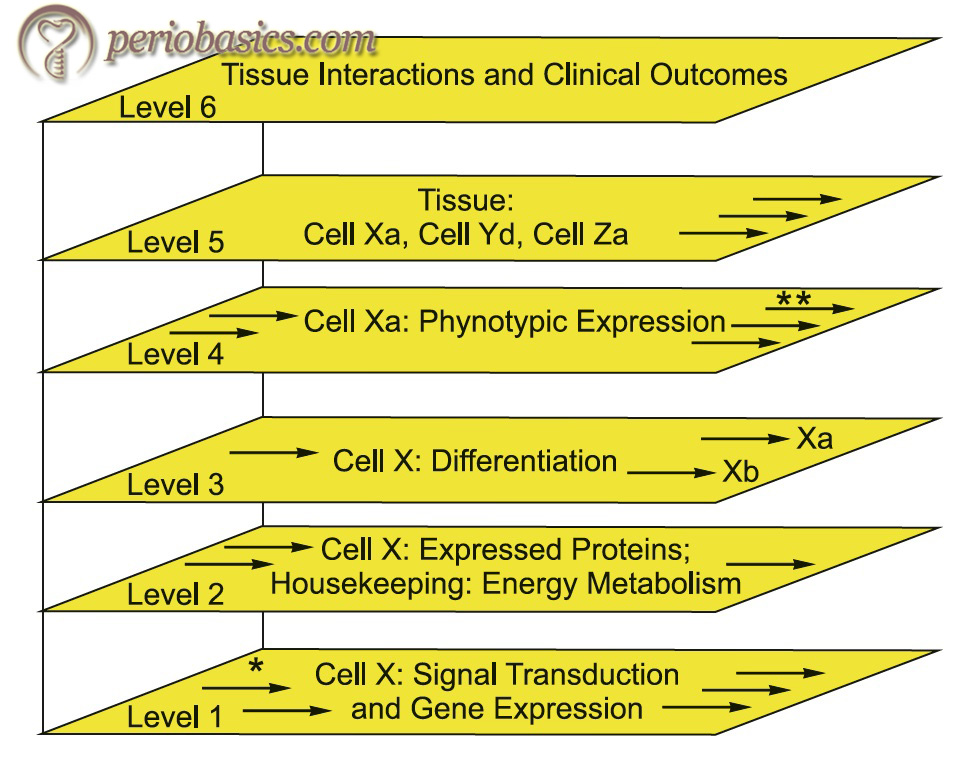
With the improvement in our knowledge of etiopathogenesis of periodontal diseases, newer concepts have been put forward. Kornman in 2008 28, put forward the multilevel hierarchical model and incorporated the newer evidence regarding periodontal disease progression in this model. He stated that the newer model should be based on a multilevel hierarchical organization, and the interactions are reflected in the gene, protein, and metabolite expression patterns. According to this model, the disease progression can be explained on the basis of ……. Contents available in the book………… Contents available in the book………… Contents available in the book………… Contents available in the book…..
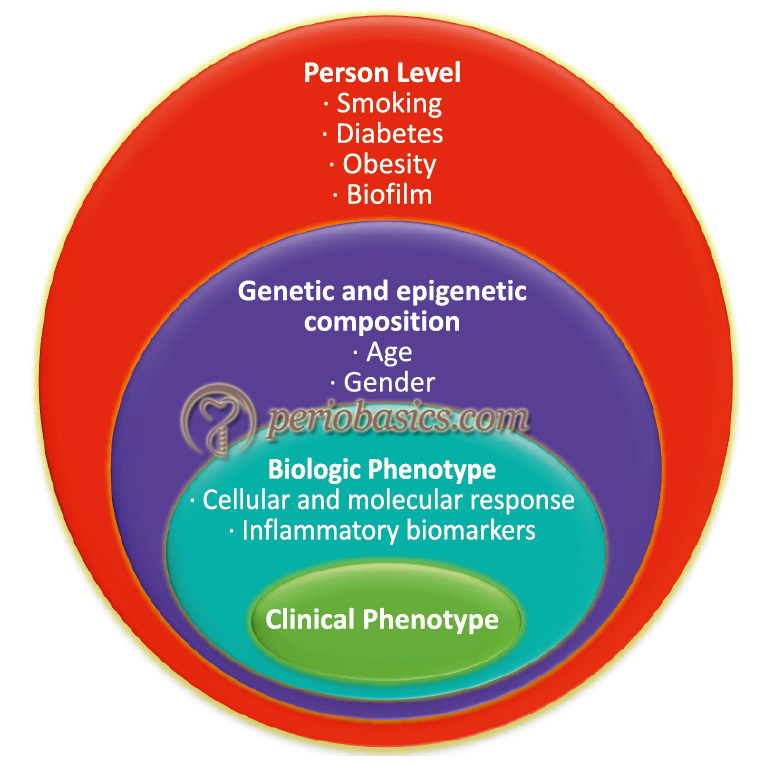
Biologic systems model by Offenbacher
Offenbacher et al. in the same year (2008) 141, proposed “biologic systems model’’ of periodontal disease progression. This model outlines the role of different components which may lead to the clinical presentation of the disease. The authors described this model with the help of four circles (Figure 7.10). The outermost circle represents the exposure at a subject level, where factors like the infecting organism and the medical history, habit history of the individual play a role. These components may interact with the next level of components which is depicted by genetics. The third and fourth circle represent the biological and clinical phenotype, respectively. The level of biologic phenotype mainly includes various biochemical markers which may affect the clinical phenotype of the patient. Thus, it can be concluded that the clinical presentation of the disease may vary from individual to individual depending on the multiple elements associated with that particular individual.
Mathematical model by Papantonopoulos
Papantonopoulos et al. (2013) 142 proposed the mathematical model of periodontal disease progression where the authors hypothesized that periodontitis is a nonlinear chaotic disease. They used clinical and immunologic records from patients with periodontitis. The mathematical model used by the authors could predict the two known types of periodontitis, aggressive and chronic periodontitis. The results of the study demonstrated that the host immune response plays a key role in periodontal disease progression. It was found that the disease progression decreased if the level of the host immune response was increased. Further, chronic and aggressive periodontitis showed a different immune response. The authors also stated that factors like genetics, smoking, and nutrition may make the system more disordered and random. The major drawback with this model is that the systemic diseases which can significantly affect the periodontal disease progression were not considered in this model.
Contemporary model of host-microbe interactions (Meyle and Chapple)
Meyle and Chapple (2015) 143 recently proposed a model of periodontal disease progression, which has evolved from the classical model of Page and Kornman27. According to this model, in clinical health, there are mild inflammatory changes that are proportionate to maintaining a resident ‘health-promoting’ periodontal microbiota. This relationship is symbiotic where the host and the microorganisms live in harmony. However, if the plaque biofilm is allowed to accumulate and is not regularly disturbed/removed, certain pathogenic bacteria are able to emerge and an ‘incipient dysbiosis’ develops. In certain susceptible patients, incipient dysbiosis can trigger ……. Contents available in the book………… Contents available in the book………… Contents available in the book………… Contents available in the book…..
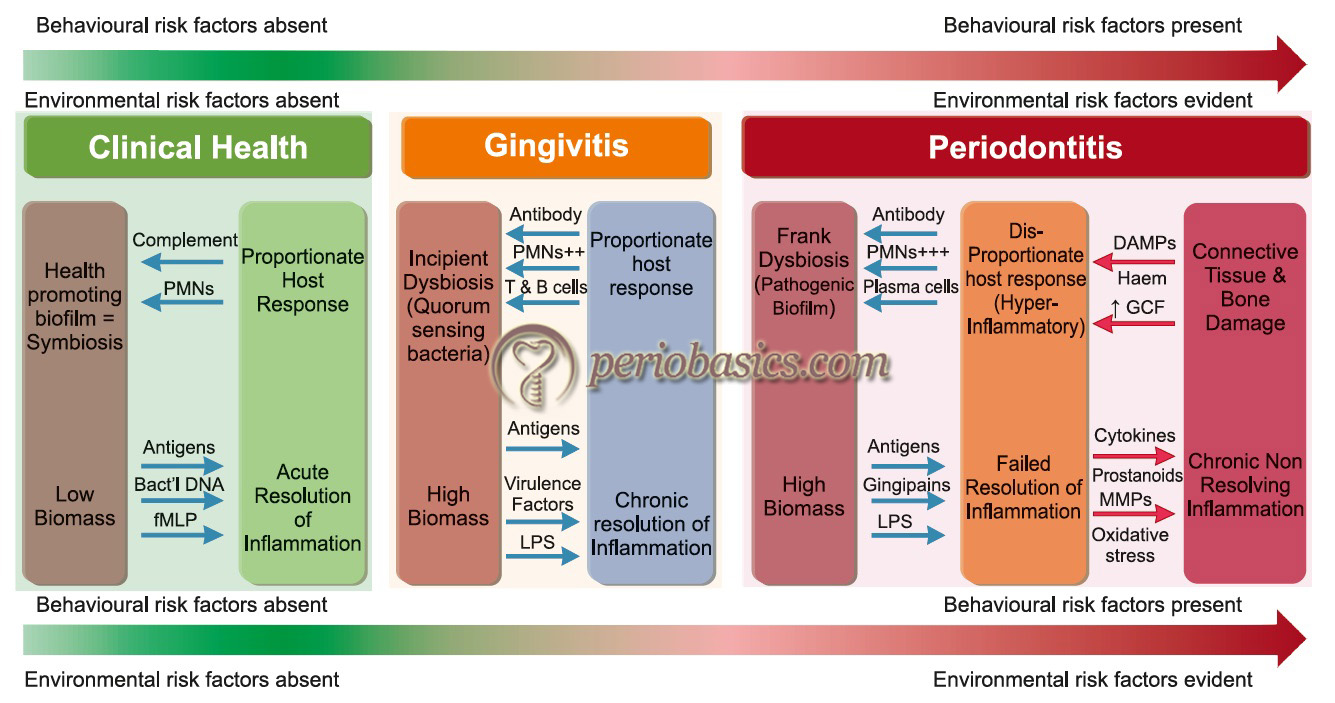
The elimination of biofilm at this point results in the re-establishment of health-promoting microbial species. The pro-resolution pathways of inflammation resolve the inflammatory process and the normal tissue structure and function of tissues are restored. However, the degree of biofilm reduction necessary to re-establish symbiosis appears to vary from person to person. If the biofilm persists, the connective tissue and bone damage occurs resulting in the progression of periodontitis. It should be noted here that if a normal distribution curve of periodontitis patients is drawn, at extreme ends of this curve, we have disease-resistant people who are at low or even zero risk of developing periodontitis (disease resistance), and high-risk patients in whom only mild biofilm accumulation appears to exceed the threshold necessary to trigger a destructive host response and subsequent tissue damage.
Our current understanding of the etiopathogenesis of periodontal diseases
It is a well-established fact that out of the bacteria species present in dental plaque, only specific bacterial species are able to cause periodontal destruction, which recently have been referred to as ‘keystone pathogens’ 144. From the above discussion, it is clear that our immune system is equipped with multiple mechanisms to counter the infections. In complete periodontal health, there is a predominance of the commensal microbiota, which is helpful in the maintenance of periodontal health. However, in a microbial community, both symbiotic and antagonistic microbial interactions are seen. In a recent hypothesis, the ‘microbial dysbiosis’ has been proposed as the basis of the etiopathogenesis of periodontal diseases. Let us now discuss this hypothesis in detail.
Role of polymicrobial dysbiosis in the etiopathogenesis of periodontal diseases
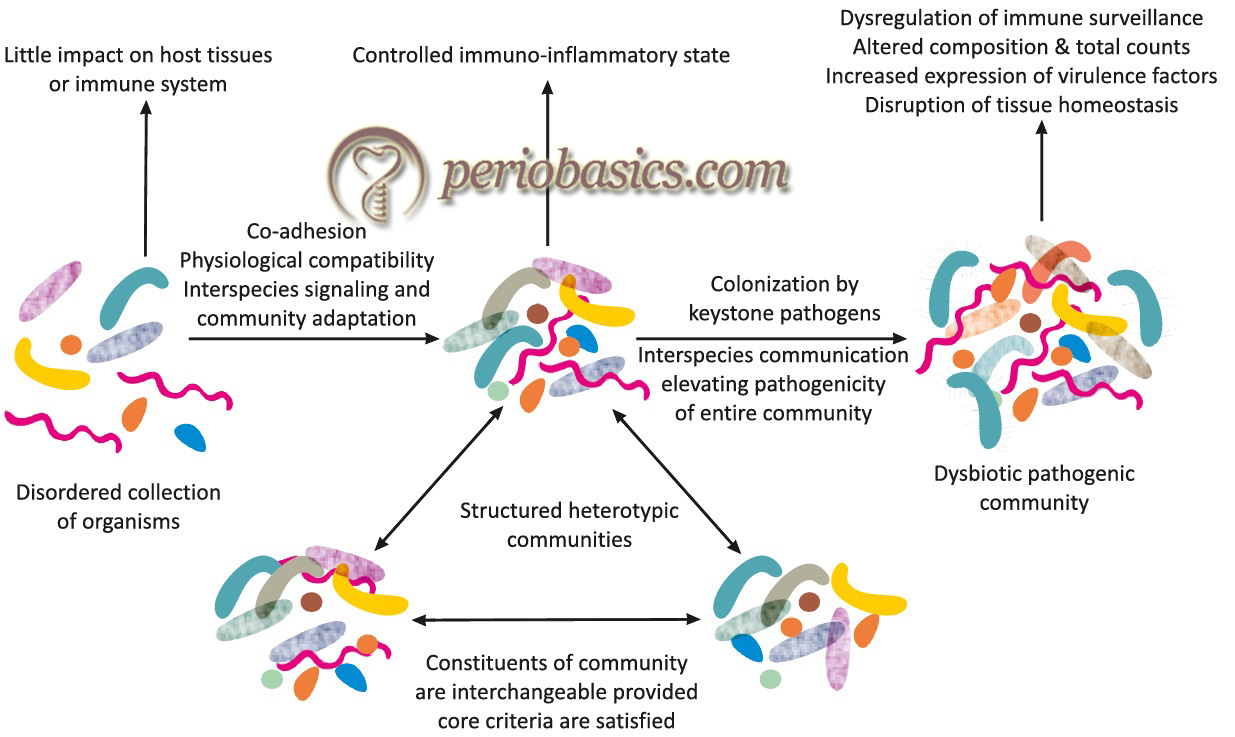
In periodontal health, the microbial communities in plaque biofilm are in a symbiotic relationship. However, when this ‘symbiotic’ relationship becomes ‘dysbiotic’, periodontal disease progression may occur. The recent hypothesis regarding the etiopathogenesis of periodontal diseases put forward by Hajishengallis and Lamont (2012) 145 is based on the concept of polymicrobial dysbiosis in a biofilm. The authors have named this model as “polymicrobial synergy and dysbiosis (PSD) model”. According to this model, in the healthy periodontium, there is a state of physiological compatibility between different microbial species which is controlled by host response. However, when this synergistic relationship is lost between the microorganisms, dysbiosis occurs in a biofilm because of which the pathogenic bacterial species proliferate and cause periodontal disease. As discussed in keystone hypothesis in “Dental plaque”, Hajishengallis et al. (2012) 145 proposed that certain bacteria have immune‐subversive properties, because of which, they have an ability to induce changes in their surrounding microenvironment that not only facilitates their own growth but also the growth of other virulent bacterial species. These keystone pathogens are the major players ……. Contents available in the book………… Contents available in the book………… Contents available in the book………… Contents available in the book…..
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
According to Hajishengallis (2014) 151, certain bacteria demonstrate an inflammophilic character, that is, they are attracted towards the site of inflammation. The inflammatory changes in tissue act as a source of nutrients for microorganisms, thus can exert a powerful influence in the composition and numbers of the periodontal microbiota. Various connective tissue degradation products such as amino acids, fatty acids or hemoglobin breakdown products such as (haptoglobin, hemopexin, and hemoglobin) act as a source of nutrition for these periodontal pathogens. Because of inflammation, there is an increased flow of gingival crevicular fluid, which further acts as a source of nutrition for these periodontal pathogens. The inflammatory changes cause alterations in the sub-gingival environment facilitating the growth of obligately anaerobic bacteria. In other words, we can say that inflammation can alter the microbial composition of the subgingival microbial community.
But, does this mean that anti-inflammatory interventions can control the periodontal disease progression? Many experimental studies have been done to find out the answer to this question. An experimental study on rabbit model, (Hasturk et al., 2007 152) demonstrated that application of resolvin E1, an anti-inflammatory mediator, not only resolved the P. gingivalis-induced periodontitis but also diminished the numbers of P. gingivalis and other Gram-negative periodontal bacteria 152. In another experiment, Eskan et al. (2012) 153, demonstrated that application of meloxicam (a selective inhibitor of cyclooxygenase-2) in sites with ligature-induced periodontitis, not only reduced inflammation but also significantly reduced bacterial load. Hajishengallis et al. (2011) 146 demonstrated that ……. Contents available in the book………… Contents available in the book………… Contents available in the book………… Contents available in the book…..
Concept of genetic dysbiosis
Recently, the concept of genetic dysbiosis has been proposed to explain the pathogenesis of periodontal diseases by Stein and Karydis (2015) 155. According to this concept, periodontal disease progression is the result of genetic defects in the recognition and response pathways that the host uses to identify microbial pathogens resulting in either altered microbial colonization or misrecognition of normal microbiota. This concept is the cornerstone of “infecto-genomics” 156. Two distinct pathways have been described under infectogenomics,
Pathogen recognition
As already explained, there is a variety of toll-like receptors (TLRs) in our body that identify pathogen-associated molecular patterns (PAMPs). Along with TLRs, other receptors that identify pathogens include NOD-like receptors (NLRs), RIG-I-like receptors (RLRs), C-type-lectin like receptors (CLRs), scavenger receptors (SCs), innate DNA receptor proteins termed AIM2-like receptors (ALRs), members of the complement pathways and peptidoglycan-recognition proteins (PRPs). These all receptors are associated with the cell membrane. However, there are many soluble pathogen recognition receptors including collectins, ficolins, pentraxins, galectins, CDs (cluster of differentiation) 14 and natural IgM (immunoglobulin M) 157-159. The binding of PAMP with any of these host-associated receptors results in a cascade of cellular signaling resulting in the generation of pro- and/or anti-inflammatory proteins such as cytokines. Any mutation in the promoter region or in the coding sequence of pathogen recognition receptor may result in their altered expression or their diminished ability to recognize PAMPs, respectively.
Bacterial proliferation
As discussed earlier in the PSD model, majority of putative periodontal pathogens have an ‘inflammophilic’ nature, that is, they prefer to proliferate at sites with inflammation. According to this hypothesis, genetic variations predispose to the generation of excessive inflammatory response that creates a favorable environment for the selective proliferation of specific putative bacteria within the biofilm. These bacterial species due to specific metabolic characteristics further create a favorable environment that favors not only their own growth but also facilitates the growth of other putative periodontopathogens.
So, we can conclude that according to the concept of genetic dysbiosis, genetic predisposition is a very important factor in the pathogenesis of periodontal diseases in susceptible individuals. However, it should be remembered that these recently proposed models of periodontal disease progression need further research work to find out their actual authenticity in periodontal disease progression.
Conclusion
Periodontal diseases are inflammatory diseases where virulent microbial species induce a series of host response events that results in the clinically visible inflammatory changes in the periodontal tissues. In the process of host-microbial interaction, there is a damage to the host tissue, which is clinically manifested as gingivitis, which may, later on, give rise to chronic or aggressive periodontitis. In a susceptible host, there is dysregulation of inflammatory and immune pathways which may result in excessive damage to the host tissues. The above discussion not only provides us the information regarding various aspects of host response, but also about the potential therapeutic approaches which can be adopted to halt the progression of the periodontal disease.
References
References are available in the hard-copy of the website.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

