Introduction to bone grafts
Bone grafting procedure involves the placement of a bone graft in the periodontal bone defect to achieve regeneration of the lost bone volume. In orthopedics, bone grafts have been used for years. The first recorded bone implant was performed in 1668. It was Hegedus 1, who in 1923 attempted the use of bone grafts for the reconstruction of bone defects produced by periodontal disease. After this, many researchers used different forms of bone grafts in periodontal bone defects. Today, many different types of bone grafts are available commercially for clinical use. A detailed description of the historical aspect of bone grafts is available in “History of periodontal regenerative therapy”.
Before we read in detail about bone grafts, the following terms should be clearly understood,
Osteogenesis:
In this case, viable osteoblasts within the grafted material deposit new bone. It occurs when autogenous bone grafts are used.
Osteoconduction:
Osteoconduction occurs when the bone graft material serves as a scaffold for new bone growth that is perpetuated by the native bone.
Osteoinduction:
It is a process in which new bone is induced to form through the action of factors contained within the grafted bone, such as proteins or growth factors.
Osteopromotion:
It occurs when the grafted material does not possess the ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Bone fill:
Bone fill is the presence of hard tissue in a periodontal osseous defect.
Autograft:
These are bone grafts harvested from patient’s own body. These are considered ideal because they possess osteoconductive and osteoinductive properties and also act as a source of osteoprogenitor cells.
Isograft:
The isograft is material that is taken from one individual and transplanted into another genetically identical individual, such as an identical twin. In isograft cases, the donor and the recipient must have the same genotype.
Allograft:
Allografts are derived from other individuals of the same species. Examples include freeze-dried bone allografts (FDBA) and demineralized freeze-dried bone allograft (DFDBA). These are the most commonly used grafting materials.
Xenograft:
Xenografts are derived from nonhuman animal sources such as bovine. The anorganic bovine bone is chemically treated to remove its organic components, leaving a trabecular and porous architecture similar to human bone.
Alloplast:
Alloplasts are synthetic bone substitutes. These are made up of hydroxyapatite, a naturally occurring mineral that is also the main mineral component of bone. Bioactive glasses are the commonly used alloplasts.
Ideal properties of a bone graft
There are some basic requirements for a bone grafting material to be designated as ideal. Every grafting material has its own advantages and disadvantages. The ideal grafting material should possess the following properties 3,
- Should be biocompatible and non-immunogenic.
- Should have no risk of disease transmission.
- Should be physiologically stable.
- Should be osteoconductive.
- Should be osteoinductive.
- Should have the ability to osseointegrate i.e., the ability to chemically bond to the surface of bone without an intervening layer of fibrous tissue.
- Should induce osteogenesis.
- Should have minimal post-operative sequelae.
- Should be cost effective.
- Should be acceptable by the patient.
Autografts are considered to be closest to an ideal grafting material because they possess all the above-stated properties. Allografts have osseointegrative and osteoconductive properties and may also exhibit osteoinductive potential, but they are not osteogenic because they do not contain viable cells. Alloplasts or synthetic graft materials possess only osseointegrative and osteoconductive properties. The following figure demonstrates the essential elements for new bone formation.

Classification of bone grafts
Based on the source of procurement, grafts have been divided into following categories 4,
Autogenous bone grafts.
Allograft.
Xenogenic bone grafts.
Synthetic/Alloplastic bone grafts.
Composite grafts.
The following figure describes the classification of various bone grafts on the basis of their sources of procurement,
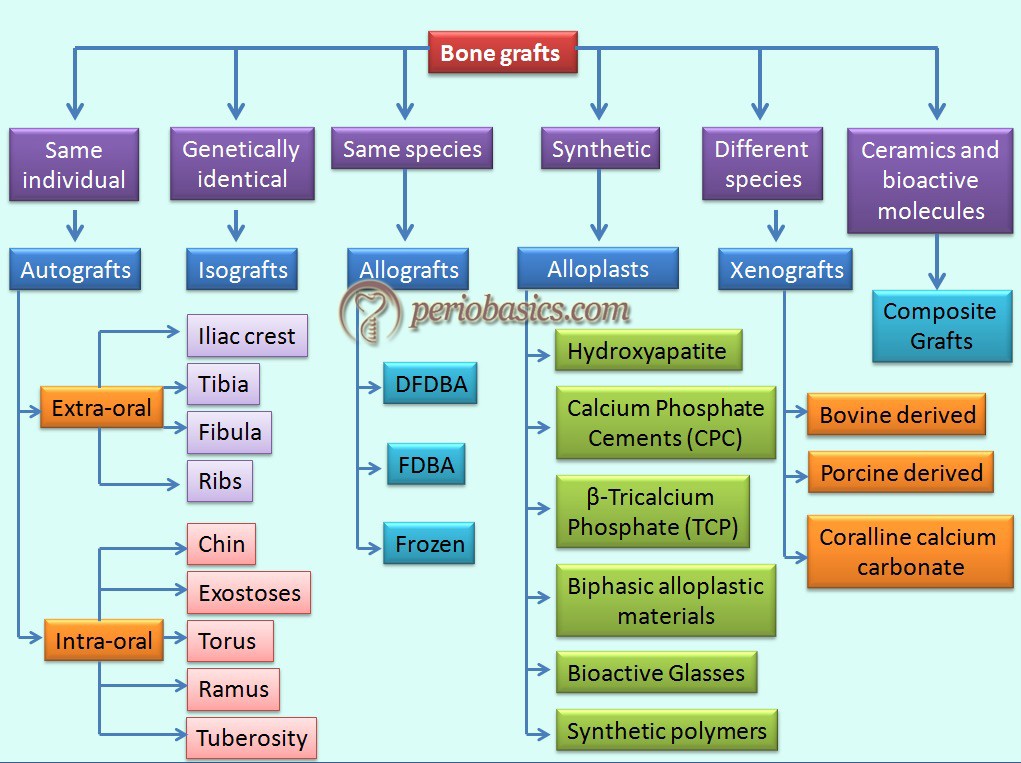
Autogenous Bone Graft
These bone grafts are obtained from the same individual on whom the bone grafting has to be performed. They may be obtained from a local/remote location. As they possess most of the properties of an ideal graft material, they are considered as the gold standard bone replacement graft. The graft obtained has viable cells, which participate in new bone formation hence has osteogenic properties. Major advantages of using autogenous grafts are: these grafts are osteogenic and there is no risk of disease transmission. Major disadvantages of autografts include procurement morbidity, limited availability in case of intraoral sites and the high cost of the procedure in case of extraoral graft harvesting.
Autografts are subdivided into three groups: cancellous, cortical and or cortico- cancellous autografts. The cancellous bone grafts are derived from the cancellous part of the bone. Because of the cancellous nature of the graft, most number of viable cells is available in this autograft. Although, after ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Sources:
Intraoral:
Intraoral autogenous bone grafts can be harvested from the edentulous alveolar areas, maxillary tuberosity, healing bony wound, extraction sites and mental and retromolar areas 5.
Cortical bone chips:
The use of cortical bone chips can be traced back to the work of Nabers and O’Leary (1965) 6. They used shavings of cortical bone removed by chisel during osteoplasty and ostectomy, in periodontal bone defects. This type of autogenous bone graft did not get much popularity and is rarely used today because of the large size (1,559.6 × 183 μm) of the graft particles, which have more vulnerability for sequestration 7.
Osseous coagulum:
The rationale behind the use of osseous coagulum and bone blend was that smaller the size of graft particles, more are the chances for its resorption and replacement with host bone 8. The bone particles were harvested by using round burs and were then mixed with blood. The rationale for mixing blood with graft particles was to provide osteogenic progenitor cells and morphogens to the wound site and to promote a stable clot formation. Studies on monkey model demonstrated that a small particle size ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Bone blend:
The bone blend procedure involves the collection of cortical or cancellous bone with the help of trephine or rongeur. Then it is put in an amalgam capsule and triturated to achieve the consistency of a slushy osseous mass. The resultant particle size is in the range of 210 x 105 μm 10. Research reports have shown clinically significant bone fill in the areas treated with bone blend 8, 11.
Extraoral:
Extraoral bone graft harvesting is a popular procedure, especially in cases where a large amount of bone graft is required. Many authors believe that extraoral cancellous bone and marrow grafts have the greatest potential for new bone formation 16-18. The most suitable site for extraoral bone graft harvesting is the iliac crest. According to Rosen et al. (2000) 19, autografts ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
In the majority of cases, a large amount of bone graft is not required, making intraoral bone graft harvesting more preferable.
Allografts
An allograft is a graft obtained from genetically dissimilar members of the same species. Allografts are obtained under sterile conditions from fresh cadavers, usually within 24 hours of the death of the donor. Their major advantages are unlimited availability and osteoinductive potential comparable to autogenous bone. These are primarily available in two forms: freeze-dried bone allograft (FDBA) and decalcified freeze-dried bone allograft (DFDBA).
So, what is the need for decalcify/demineralize a cortical bone allograft? Demineralization of the cortical bone allograft improves the osteoinductive potential by ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… There are set standards for procuring, processing and sterilization of allografts (FDBA/ DFDBA). Most of the bone banks adhere to the guidelines of the American Association of Tissue Banks (AATB), as described by the Centers for Disease Control and Prevention 20. According to AATB, allografts should not be collected if,
1. The donor is from high-risk groups, as determined by medical testing and behavioral risk assessments.
2. Donor has been tested positive for HIV antibody by ELISA.
3. Autopsy of donor reveals occult disease.
4. Donor’s bone has been tested positive for bacterial contamination.
5. Donor and bone have been tested positive for hepatitis B surface antigen (HBsAG) or hepatitis C virus (HCV).
6. Donor has been tested positive for syphilis.
Processing of allografts:
Although allograft manufacturing companies do not disclose the exact method of bone processing they follow, however following is the description of the basic technique of bone processing,
The first and foremost step is obtaining the bone from a suitable donor and reducing it into pieces of small size of approximately 5 mm. The second step is the elimination of bone marrow and cellular debris. The elimination of bone marrow and cellular debris is achieved with fluids and detergents, which, by their clearing effect, improve the osteoconductive capacity of the bone. Pressurization allows full penetration of inactivating or eliminating agents into the bone. Various chemical solutions used in this procedure include saline, acetone, ethanol or hydrogen peroxide which remove bioburden and reduce antigenicity. Then bone particles are treated with antimicrobial, antimycotic and antifungal solutions. After this, bone particles are kept in ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
For preparing DFDBA, the decalcification process follows after final particle size of 250 to 750 μm is achieved. The bone particles are immersed in a hydrochloric acid bath at concentrations ranging from 0.5 to 0.6 N for various durations of time. Then these acid treated particles are immersed in a buffering solution to remove residual acid. The demineralized allograft is further rinsed with various solutions (e.g., distilled water) to remove the residual buffer solution. Then graft is packed in sterile containers followed by irradiation with low-dose of γ radiations at low temperatures to ensure sterility.
Bone graft processing results in an exponential reduction in graft contamination and disease transfer or both. Sterility assurance level (SAL) is a term used to describe the probability that an item will not be sterile ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
After processing bone allograft has to undergo certain tests which include:
Visual inspection test:
Visual detection is done for problems such as gross graft contamination, packaging defects and product mislabeling.
Residual moisture test:
Testing of FDBA is done to ensure that the residual moisture is 6 percent or less.
Residual calcium test:
Testing of DFDBA is done to ensure that the residual calcium content is 8% or less.
Once the allograft has passed all the above-stated tests, it is packaged and sent for the clinical usage. There are various commercially available allografts, some of which are discussed below,
Commercially available allografts
Puros (Zimmer Dental, Carlsbad, California):
This is a commercially available allograft obtained from human source. It is subjected to patented tutoplast process which gently removes the undesirable components of the bone including fats, cells, antigens, and inactivates pathogens while preserving the valuable minerals and collagen matrix, leading to complete and rapid bone regeneration 23. Various chemicals are used to eliminate these unwanted components, including acetone to remove lipids, H2O2 to ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Grafton DBM (BioHorizons, Birmingham, Alabama):
This allograft is derived from cadaver long bones. The procured bone is subjected to chemical processing under aseptic conditions to remove unwanted bone components such as lipid, blood, and cellular components before it is frozen. The resultant bone is subjected to the reduction in particle size and stored in sterile containers. The graft is combined with a glycerol carrier to stabilize the proteins and improve the graft handling 24.
Xenografts
As already stated, xenografts are the grafts transferred between genetically dissimilar members of different species. These are derived from three sources: bovine bone, porcine bone, and natural coral. These are osteoconductive, biocompatible and similar to human bone in structure 25, 26.
Bovine-derived xenografts:
Bovine-derived xenografts are processed by removing all the organic content of the bone to minimize any chances of graft rejection. The procedure involves the application of various chemicals on the bone. The raw bone is subjected to boiling in alkalis (such as potassium hydroxide) followed by maceration in hydrogen peroxide and ethylenediamine. The resultant graft has a hydroxyapatite skeleton with porous internal structure with large surface area, which facilitates revascularization and integration into the host bone 27, 28. A porous ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Commercially available bovine-derived xenografts
Bio-Oss ® (Osteohealth Co., Shirley, NY):
This is a bovine-derived bone grafting material. The unwanted organic material within the bone is removed by chemical processing at a low temperature, resulting in a porous mineral matrix. Processing at low temperature has an advantage of preservation of natural architecture of the bone. This graft material is available in cancellous and cortical granules and blocks. The particle size of the graft is approximately 100 × 200 × 500 Å.

Bio-Oss Collagen ® (Osteohealth Co., Shirley, NY):
This graft has inorganic and organic components, consisting of Bio-Oss Spongiosa granules (0.25–1 mm) and 10% highly purified porcine collagen. The addition of collagen in the graft material improves handling characteristics of the graft without acting as a barrier. The graft particles are adhered to each other due to the presence of collagen, which facilitates graft placement without a membrane. The collagen component is resorbed within 4-6 weeks.
OsteoGraf/N ® (CeraMed Dental, LLC, Lakewood, CO):
It is a natural anorganic bovine-derived microporous hydroxyapatite. It is the only xenograft that meets all ASTM (American Society for Testing and Materials) standards for “Composition of Anorganic Bone for Surgical Implants (F1581-95). It achieves a hydrophilic – cohesive consistency when hydrated. It remodels to the vital bone at the same rate as the host bone. It is manufactured as radiopaque, rounded particles and is available in two particle sizes:
OsteoGraf/N-300 (250-420 μm) packaged in 1 gram and 3 gram vials.
OsteoGraf/N-700 (420-1,000 μm) packaged in 1 gram and 3 gram vials.
PepGen P-15 ® (Dentsply Friadent, Mannheim, Germany):
It is a tissue-engineered bone replacement graft material which mimics the inorganic and organic components of autogenous bone. It consists of a uniquely designed P-15 peptide, a synthetic biomimetic of the 15 amino acid sequence of collagen Type-I, which is uniquely involved in the binding of cells, particularly ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Porcine-derived Xenografts:
Xenografts derived from porcine cortical and cancellous bone have also been developed to be used as bone substitutes.
OsteoBiol ® Gen-Os (Tecnoss Dental, Turin, Italy):
It is commercially available xenograft of porcine origin. It is a heterologous cortico-cancellous collagenated bone mix. It must always be hydrated before use. It is thoroughly mixed with a few drops of sterile physiological solution to activate its collagen matrix and to enhance its adhesivity. It can also be mixed either with OsteoBiol Gel or with the patient’s blood. A major advantage of this material is that it can act as a carrier for various therapeutic agents and drugs. The collagen present in this ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Coralline calcium carbonate:
These bone grafts are derived from natural coral. The structural features of commercially used coral and Porites are similar to that of cancellous bone, which makes them a suitable bone substitute.
Biocoral ® (Inoteb, Saint Gonnery, France) is a commercially available grafting material derived from natural coral which has a calcium carbonate structure primarily composed of aragonite. It has a porous structure with a pore size of 100 to 200 μm. This porous structure provides a large surface area, helping in a better cellular resorption and replacement by natural bone. Its osteoconductive properties are comparable to other bone substitutes.
Alloplasts
Alloplasts are synthetic bone substitutes that are readily available and also eliminate the need for a patient donor site. Like other bone grafting materials, the ideal properties of alloplastic materials include: it should be biocompatible with host tissues, non-antigenic, non-carcinogenic, and non-inflammatory. Along with this, the graft material,
- Should be sufficiently porous and should allow tissues to grow into and around the implant (osteoconduction),
- Should be able to stimulate bone induction, should be resorbable and replaceable by bone,
- Should be radio-opaque in order to be visualized radiographically,
- Should be able to withstand sterilization without losing favorable qualities,
- Should be stable at varying temperatures and humidity,
- Should have surface electrical activity (i.e., be charged negatively),
- Should be hydrophilic,
- Should be easy to manipulate clinically,
- Should have high compressive strength, and
- Should be inexpensive, and easily attainable.
Various synthetic graft materials include hydroxyapatite, tricalcium phosphate, calcium sulfate (plaster of Paris), bioactive glasses, and hard tissue replacement polymers. Following is a detailed description of these materials,
Hydroxyapatite (Ca4 (PO4)6(OH)2):
Hydroxyapatite (HA) is the main component of inorganic components of bone. HA ceramics have a stoichiometry similar to that of bone mineral 29, 30. It can be manufactured from natural reef-building coral skeleton by a “hydrothermal exchange reaction”, where the trabecular, bone imitating structure of the coral remains unchanged and the calcium carbonate (CC) skeleton is converted into calcium phosphate, the main inorganic salt of bone 31. The main difference between calcium carbonate and HA is markedly slow resorption of HA as compared to calcium carbonate. Biodegradation should not occur before the graft implant is filled with bone. The slow resorption of HA allows the formation of new bone in the graft due to its porosity. HA lacks the capacity to induce bone growth.
Other methods of manufacturing porous HA include homogenizing calcium phosphate powder with appropriately sized naphthalene particles, resulting in macroporous material after ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Depending on the method of procurement and processing, HA grafts are available in following forms,
Polycrystalline ceramic form of pure densely sintered HA:
It is a polycrystalline form of HA prepared in relatively large particle size (18-40 mesh). Calcitite, a commercially available HA graft (Calcitek, Inc., Carlsbad, CA), has a particle size of 420-840 μm. OsteoGraf/D300 and OsteoGraf/D700 (CeraMed Corp., Lakewood, CO), have particle sizes 250-420 μm and 420-1,000 μm respectively.
Coralline porous non-resorbable hydroxylapatite:
As already stated they are derived from the marine coral skeleton from which organic components have been removed. Interpore 200 (Interpore International, Irvine, CA) and Pro-Osteon 500R (Interpore Cross International, Irvine, CA, USA) are the examples of commercially available coralline porous non-resorbable hydroxylapatite.
Resorbable nonceramic hydroxylapatite:
It is a resorbable form of HA which is highly microporous and non-sintered (nonceramic). Particle size varies from 300-400 μm. Commercially available examples of this type of HA grafts include OsteoGen® (Impladent, Holliswood, NY) which is a low-temperature hydroxylapatite (HA) material. It is manufactured by processing at low temperature and is not sintered. These non-sintered precipitated particles have size measuring from 300-400 μm. Other examples include OsteoGraf/LD-300® (particles are sized between 250 and 420 μm) (CeraMed Corp., Lakewood, CO) and Cerabone ® (Coripharm GmbH & Co. KG, Dieburg, Germany).
Nanocrystalline hydroxyapatite (NHA):
As already stated, hydroxyapatite is a naturally occurring mineral form of calcium apatite. It comprises about 50% of the weight of the bone. The material has excellent osteoconductive and osteointegrative properties. Porous HA has similar initial mechanical properties compared to the cancellous bone but is brittle and weak under tension and shear forces. Its resistance to compressive loads decreases by 30-40% in situ after being implanted for several months. The macro-porosity of the HA graft material (pore with diameters > 100 mm), as well as the interconnectivity between the pores, allows the ingrowth of osteoprogenitor cells, as well as the revascularization thereby facilitating the ingrowth of new bone. However, because of ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
As the name indicates, NHA bone graft has nano-sized particles. This graft material has larger surface to volume ratio. The great surface not only significantly reduced the sintering temperature of HA ceramic, but also leads to the increased resorption rate. Hence, the main advantage of this biomaterial is improved osteoconductive properties and complete resorption of the material within 12 weeks. A commercially available example of this graft is Ostim™ (Heraeus Kulzer, Hanau, Germany) (NHA), which is a synthe-tic nanocrystalline hydroxyapatite (NHA) paste containing 65% water and 35% nanostructured apatite particles.
Another advancement in NHA graft materials is the incorporation of carbon nanotubes (CNTs). It not ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Fluorohydroxyapatitic (FHA) biomaterials:
Historically, it was observed that some forms of calcified algae have an internal structure similar to cancellous bone 33. So, its potential use as a bone substitute was proposed.
FRIOS ® Algipore ® (Friadent GmbH, Mannheim, Germ-any) is a commercially available FHA bone graft. It is derived from calcifying marine algae (Corallina Officinalis). This material has got micro-perforations which are inter-connected. The average diameter of the pores is around 10 μm and every pore is limited by one layer of small FHA crystallites with a size of 25-35 nm. The material has good osteoconductivity.
β-Tricalcium Phosphate:
Tricalcium Phosphate (TCP) is present in two forms: α and β. The α-TCP is monoclinic and consists of columns of cations, while the β-TCP has a rhombohedral structure. The physical properties of these two forms are different. α-TCP is less stable than β-TCP and when mixed with water, it forms calcium-deficient hydroxyapatite. β-TCP has a physical structure closer to the bone, but its compressive strength is almost 1/20th of the cortical bone. The osteoconductive properties of this grafting material have been shown to be good 34, 35. After placement in the bone defect, this material is slowly resorbed and replaced by natural bone, which is facilitated by the porous structure of the material. The exact mechanism of bone graft resorption is still ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Commercially available β-Tricalcium Phosphate (TCP):
There are various commercially available β-TCP bone graft materials available for clinical application. Some of these are discussed below,
Bioresorb ® ( Sybron Implant Solutions GmbH Bremen, Germany):
It is a β-TCP bone grafting material having a porous structure similar to human bone. The manufacturers claim a micropore sizes range from 0.5 -10 μm, and the macropore sizes are 50-700 μm which provides a highly porous structure for new bone formation and slow graft resorption. The major advantage of using pure β-TCP is its high osteoconductivity but at the same time it is not as stable as hydroxyapatite and is resorbed at a faster rate.
Cerasorb ® (Curasan, Kleinostheim Germany):
This synthetic graft material has a particle size of granules between 150-500 µm. It is a pure-phase β-TCP. It is completely resorbed in 4-12 months and its porous scaffold during that time helps the natural bone formation to occur. It does not contain HA, so its resorption occurs completely, uniformly and parallel to the formation of the surrounding bone. The granules are radio-opaque, resorbing gradually from the inside out, so clinicians can easily monitor the status of grafted site.
Vitoss ® (Orthovita, Malvern, PA, USA) :
It is a β-TCP synthetic bone substitute. The material is 3-dimensionally macroporous, containing spaces into which bone ingrowth takes place. It does not have significant compressive strength by itself.
Calcium Phosphate Cement (CPC):
CPC utilizes two phases of components, one in powder form and another in liquid form. The powder is usually a mix of different calcium phosphate salts where almost all products contain one or several of the components, including amorphous calcium phosphate (ACP), dicalcium phosphate dihydrate (DCPD), dicalcium phosphate anhydrous (DCPA), α-tricalcium phosphate (α-TCP), dicalcium phosphate (DCP), tetra-calcium phosphate (TTCP), monocalcium phosphate monohydrate (MCPM) and calcium carbonate (CC). The liquid component usually contains sodium phosphate solution. When mixed, the material sets to form a solid mass. After mixing the material, a workable consistency of the material is achieved and the bony defect is filled and contoured.
The porosity of the set mass can vary between 30% and 50%, depending on the processing conditions, e.g. liquid-to-powder ratio. The setting, rheological and mechanical properties of the CPC can be adjusted by modifying different factors, such as the chemical composition of reactants, particle size or presence of nucleating agents 40-43. Various ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
The problems associated with these materials are prolonged setting time and inability to set in the presence of blood. To overcome these problems new improved cements have been developed. Following are the commercially available calcium phosphate cements used in periodontal bone defects,
Norian® PDC™ (Shofu Inc., Kyoto, Japan):
It is an injectable, moldable, fast setting, bioabsorbable calcium phosphate cement having a high compressive strength (55 MPa). The powder of the cement consists of α-TCP (α-Ca3[PO4]2), monocalcium phosphate monohydrate (Ca[H2PO4]2·H2O) and calcium carbonate (CaCO3). The liquid contains a solution of sodium phosphate. The pore diameter of the set mass is around 300 Å.
Augmentech AT (Wetzlar, Germany):
This is also a moldable, fast setting, bioabsorbable cement having high compressive strength. The powder of the mix contains TCP, magnesium phosphate, magnesium hydrogen phosphate and strontium carbonate. The liquid is a watery solution of diammonium hydrogen phosphate.
Calcibon ® (Biomet):
It is a synthetic, biodegradable, calcium phosphate based bone substitute. The powder of the mix contains Powder: α-TCP (61%), DCP (26%), CaCO3(10%), and PHA (3%). The liquid contains H2O and Na2HPO4. The set material has got high compressive strength (up to 60 MPa). Compressive strength increases during setting of the material. After 6 hours, it is comparable to cancellous bone. The final compressive strength is reached after 3 days.
Bioactive Glasses:
These are silicate-based alloplasts containing calcium and phosphate. Hench (2006) 48 was the first one to develop a bioactive glass that could bind to the tissue. The formation of carbonated hydroxyapatite layer on its surface, when placed in body fluids, makes this material bioactive. The FDA approved composition of bioactive glass is designated as 45S5. The 45S5 bioactive glass is composed of SiO2 (46.1 mol%),CaO (26.9 mol%), Na2O (24.4 mol%) and P2O5 (2.6 mol %) 49. The high amounts of Na2O and CaO, as well as the relatively high CaO/P2O5 ratio, make the glass surface highly reactive in physiological environments 50. 45S5 is able to form HCAP (hydroxycarbonated apatite) in less than 2 hours and binds to the tissues. The bioactive glasses are obtained basically by two processes: melting and sol-gel process. The sol-gel process is a preferred process because it requires lower temperature as compared to the conventional melting process. It has also been reported that glasses made from sol-gel technique have increased bioactivity 51.
There are two types of responses generated when an alloplast is implanted in the body: biochemical and cellular. The biochemical response is generated due to the changes in the environment around the alloplast. When bioactive glasses are placed in vivo, a highly basic environment with pH around 10 is created, because of which a layer rich in silica gel is formed on the surface of glass particles. Because of interaction with surrounding fluid, a layer of calcium phosphate is formed which is made up of hydroxycarbonate apatite (HCA). Hydroxycarbonate apatite is chemically and structurally very similar to the bone. The cellular reactions include colonization, proliferation, and differentiation of relevant (bone) cells 50, 52.
With the development in materials over the years, new bioactive glass compositions have been introduced. These materials contain no sodium or have additional ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Commercially available bioactive glass alloplasts
PerioGlas ® (Block Drug Co., NJ, USA):
PerioGlas is a synthetic absorbable osteoconductive bone graft substitute, composed of a calcium phospho-silicate bioactive glass, Bioglass. This graft material is available for use since 1995. The particle size of the graft particles varies from 90-710 μm. It is supplied sterile, packaged either in a Tyvek-sealed PET-G cup or in a filled syringe within a second sterile barrier package. While using, the graft is mixed with sterile water, saline, patient’s own blood or marrow or with autogenous or allograft bone to form a wet sandy paste that is applied to the defect 69.

PerioGlas ® Plus (Block Drug Co., NJ, USA):
It is composed of a calcium phosphosilicate material and a calcium sulfate binder. The inorganic calcium and phosphorous components are thermally incorporated in a sodium silicate network (PerioGlas®), designed specifically for its absorbability and osteoconductive nature. The calcium sulfate incorporated in the graft material binds the bioactive glass particles and is absorbed slowly. The calcium sulfate in the PerioGlas® Plus is absorbed between 4 to 8 weeks after implantation, depending on the graft site, size and material used. The remaining PerioGlas® particle left behind, are also absorbed and slowly replaced with host bone, usually within six months 70.
Unigraft ® (Unicare Biomedical Inc., Laguna Hills, CA, USA):
Unigraft is made up of synthetic bioactive glass material. The graft material is composed of fused oxides of calcium, phosphorus, silicon and sodium. Upon implantation, the material begins to dissolve by gradually releasing a steady stream of Ca and P ions, along with soluble silica into the bony defect. This increased concentration of local bone mineral ions has been demonstrated to enhance bone regeneration and exhibit an anti-bacterial effect. The particle size of the graft material varies from 200 μm to 420 μm. It is supplied sterile in foil-sealed polyolefin vial. The graft material is mixed with sterile saline or with patient’s blood to form a sandy paste, that is applied to the defect 71.
Biogran™ (Orthovita Inc., Malvern, PA, USA):
Biogran is a synthetic bone graft material consisting of 300-355 μm diameter bioactive glass granules. It is composed of bioactive salts of Si, Ca, Na, P. The graft material can be mixed with the patient’s blood or sterile saline within the disposable dappen dish cup and subsequently delivered to the defect site. With due course of time, it is slowly resorbed and replaced by host bone.
Biocompatible osteoconductive polymers:
These are nonresorbable, particulate of calcium layered with polymethylmethacrylate and hydroxyethylmethacrylate (PMMA-PHEMA) 72. The polymers used as bone substitutes can be classified as natural and synthetic polymers which can further be divided as degradable and nondegradable 73. Present biocompatible polymers contain calcium hydroxide and polymethylmethacrylate (PMMA) and polyhydroxylethyl-methacrylate (PHEMA). The commercially available polymer graft is HTR TM Synthetic Bone (Bioplant, Norwalk, CT). This composite is prepared from a core of PMMA and PHEMA with a coating of calcium hydroxide 74. The graft material is highly porous with 150-350 μm pore size. Being polymers, these grafts are ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Composite Grafts
These are newly emerging grafting materials. Composite grafts combine scaffolding properties with biological elements to stimulate cell proliferation and differentiation and eventually osteogenesis. These contain synthetic osteoconductive matrix and osteogenic cells and growth factors which make the graft osteoinductive. Because this combination provides a scaffold as well as molecules which have osteogenic properties, these graft materials become a close replacement of autogenous bone grafts. The osteoconductive matrix becomes a carrier for the bioactive molecules. The potential combinations of graft materials for making a composite graft include bone marrow/synthetic composites, ultra porous β-TCP/BMA composite, osteoinductive growth factors and synthetic composites, BMP/polyglycolic acid polymer composites and BMA/BMP/polyglycolic acid polymer composite 29.
Commercially available composite grafts:
Healos ® (Orquest, Mountain View, CA):
Healos ® is a matrix of bovine fibrillar Type I collagen coated with HAmineral, which constitutes approximately 25% of the implant by weight. It can be mixed with bone marrow aspirate to provide osteogenic and osteoinductive potential. Another addition to this bone graft is MP52, which is a member of the BMP family. The addition of this protein further increases the osteogenic potential of this bone graft.
Collagraft ® (Zimmer Corp, Warsaw, IN):
Collagraft is a composite of suspended fibrillar collagen and a porous calcium phosphate ceramic, in a ratio of 1:1. The fibrillar collagen is highly purified ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Tricos ® (Baxter BioSciences BioSurgery):
Tricos ® is a combination of hydroxyapatite-coated beta-tricalcium phosphate (HA/TCP) granules and a fibrin matrix. This bioactive material provides a three-dimensional osteoconductive environment for the formation of new bone.

Factors affecting the success of bone grafts
Local factors:
Defect size and topography:
It is a very important factor which affects the success of bone graft. The predictability of graft success generally increases as the number of remaining bony walls increases. In other words, we can say that the graft success is most predictable with three wall and least predictable with one wall defect. A deep, narrow defect is more predictable for regeneration than a shallow and wide defect 83, 84.
Presence of infection:
The presence of infection at the grafting site is a major factor responsible for graft failure. Under low pH conditions, bone and graft material are rapidly absorbed through solution mediated resorption. Therefore, it is important to eliminate all reasons for inflammation before the placement of bone graft.
Graft stability:
The graft material should be stable at its position to facilitate a proper biological response during healing. An improperly placed graft material with vulnerability to movement is bound to fail.
Space maintenance:
The area in which regeneration has to take place requires space maintenance. If the graft material resorbs too quickly, there is no sufficient time for new bone formation and the defect gets filled with the connective tissue rather than new bone 84, 85.
Healing period:
The healing period varies from defect to defect. For a smaller three wall defect, the healing period may be shorter as compared to a large defect in which a large amount of bone graft is placed with less autogenous bone and fewer remaining walls. An adequate healing period must be given for regeneration to take place 85, 86.
Adequate blood supply:
For proper healing in an area where bone graft has been placed, an adequate blood supply is of paramount importance. The blood supply in this area is derived from two sources: the cortical or cancellous bone and soft tissue covering the defect. The cortical bone has few arterioles as compared to cancellous bone 86, 87.
Primary closure:
Primary closure of the soft tissue at the operated site is paramount for graft success. The opening of the incision line is one of the most common complications during post-operative healing. As a result of this, the graft material may be lost, contaminated and vascularization is delayed, causing graft failure.
Regional acceleratory phenomenon (RAP):
It is a local response to injury; in which healing takes place at a faster rate as compared to normal regeneration process 86. It is basically attributed to the increase in the ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ………
Effect of growth factors:
The presence of growth factors is required for regeneration to take place. These growth factors are derived from various cellular sources like platelets, macrophages and other cells involved in the local inflammatory response. Various growth factors involved are, epidermal growth factor, fibroblast growth factor, insulin-like growth factor, keratinocyte growth factor, platelet-derived growth factor, transforming growth factor, vascular endothelial growth factor, etc. A detailed description of these factors is available in “Application of growth factors in periodontal regeneration”.
Particle size of graft material:
The acceptable size of graft particles ranges from 125-1000 μm. A minimum space of 100 μm is required between the graft particle to allow vascularization and bone formation. A particle size less than 100 μm elicits macrophage resorption of the graft particles causing their early loss 7. Most commercially available bone grafts have a particle size ranging from 250-750 μm.
Systemic factors:
Systemic condition:
Systemic conditions like diabetes mellitus, hyperparathyroidism, thyrotoxicosis, osteoporosis, Paget’s disease, etc., have adverse effects on the healing of the bone graft. It must be noted here that in well-controlled diabetic patients, graft healing is similar to that in non-diabetic patients. Age is also an important factor that effects graft healing. Children heal more quickly than adults and healing potential is decreased with advancing age. The bone graft integration and quality of bone formation has been found to be poor in elderly patients as compared to adults.
Habits:
Smoking and alcohol also adversely affect the healing of the bone graft. Nicotine administration slows cartilage differentiation and results in delayed hard callus formation.
Conclusion
Bone substitutes and their application in periodontal regenerative therapy is presently a major field of research in periodontology. In the above discussion, we have discussed various bone grafts and their sources in detail. Although autogenous bone graft is closest to the ideal bone graft, but it requires an extra surgical site to harvest the graft. The allografts provide an alternative to autograft but are not as osteoinductive as autografts are. Synthetic bone substitutes such as various forms of hydroxyapatite, β-TCP, biphasic alloplastic materials, calcium phosphate cements (CPCs) and bioactive glasses have provided us a good alternative to autogenous bone grafts. As these materials have a composition which is very similar to the bone, they get resorbed and replaced by host bone in due course of time.
The future of bone graft materials is composite bone grafts, which have combined properties of the scaffold and biologically active molecules such as bone morphogenetic proteins/growth factors. In other words, we can say that the graft material acts as a carrier for biologically active molecules. Incorporation of collagen matrix into these biomaterials makes them ideal for carrying these biologically active molecules. Although the research on these materials is in its initial stage, they may be the closest replacement of autografts. The future directions in bone grafts and other aspects of periodontal regeneration have been discussed in “Tissue engineering in periodontics”.
References
References are available in the hard-copy of the website.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

