Introduction to platelet concentrates
With our improved understanding regarding cellular and molecular mechanisms involved in periodontal wound healing, various regenerative techniques including, root surface biomodification, guided tissue regeneration, guided bone regeneration and application of growth factors and platelet concentrates have been introduced in periodontal regeneration. Application of platelet concentrates has been our focus of research for quite some time now. We have a strong evidence derived from medical and dental literature supporting the use of autogenous platelet-rich plasma in clinical practice 1, 2. Platelet-rich plasma (PRP) was approved for clinical use in 1998 3. Since then there have been numerous uses of platelet concentrates including, dental implant procedures, bone grafting, periodontal surgeries and its application directly into extraction sockets to facilitate healing.
In the present discussion, we shall study in detail various forms of platelet concentrates and their application in periodontal regeneration.
Platelet function during wound healing
Wound healing is a complex process which involves many cell types and growth factors 4-6. Platelets contain high quantities of key growth factors, which are involved in cell proliferation, matrix remodeling, and angiogenesis. These include PDGF-AB (platelet-derived growth factor AB), IGF (insulin-like growth factors), TGF β-1 (transforming growth factor β-1) and VEGF (vascular endothelial growth factor), which have a well-established role during wound healing 7-9. The rationale behind the use of platelet concentrates is to ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
| Growth factors secreted by platelets and their target cells and functions | ||
|---|---|---|
| Growth factors | Target cell | Actions |
| PDGF | Fibroblasts Glial cells Smooth muscle cells Macrophages Neutrophils | · Stimulates chemotaxis/mitogenesis of fibroblasts/glial cells/smooth muscle cells. · Regulates collagen synthesis. · Stimulates macrophage/neutrophil chemotaxis. |
| TGF-β | Fibroblasts Marrow stem cells Endothelial cells Epithelial cells Pre-osteoblasts | · Stimulates/inhibits endothelial, fibroblastic, osteoblastic mitogenesis. · Regulates collagen synthesis/secretion. · Regulates mitogenic effect of other growth factors. · Stimulates endothelial chemotaxis and angiogenesis. |
| PDEGF | Endothelial cells Epithelial cells Fibroblasts | · Stimulates endothelial chemotaxis/angiogenesis. · Regulates collagen secretion. · Stimulates epithelial/mesenchymal mitogenesis. |
| PDAF | Endothelial cells | · Increases angiogenesis and vessel permeability. · Stimulates mitogenesis of endothelial cells by direct or indirect actions. · Several cytokines and growth factors up-regulate PDAF including IGF-I, TGF-α, and β, PDGF, FGF, PDEGF and IL-I β. |
| IGF-I | Osteoblasts Macrophages Monocytes Chondrocytes | · Stimulate cartilage growth, bone matrix formation and replication of pre-osteoblasts and osteoblasts. · Acts as an autocrine and paracrine factor. · In combination with PDGF can enhance the rate and quality of wound healing. |
| PF-4 | Fibroblasts Neutrophils | · Chemoattractant for fibroblasts and neutrophils. · Potent anti-heparin agent. |
| PDGF: Platelet-derived growth factor. TGF-β: Tissue growth factor-beta. PDEGF: Platelet-derived epidermal growth factor. PDAF: Platelet-derived angiogenesis factor. IGF-I: Insulin-like growth factor. PF-4: Platelet factor 4. bFGF: Basic fibroblast growth factor. |
||
Components of PRP and their role in bone regeneration
Studies on PRP have identified following important growth factors in the alpha granules of the sequestered platelets:
1. Platelet-derived growth factor (PDGF).
2. Transforming growth factor- (TGF-β).
3. Platelet-derived epidermal growth factor (PDEGF).
4. Platelet-derived angiogenesis factor (PDAF) or Vascular endothelial growth factor (VEGF).
5. Insulin-like growth factor-I (IGF-I).
6. Platelet factor-4 (PF-4).
Also, Fibrin, fibronectin, and vitronectin are present in PRP, which of course are not growth factors, but they are cell-adhesion molecules.
After injury, clot is formed which is made up of a fibrin network in which various cells like RBCs, platelets and PMNs are embedded. Platelets contain three types of secretory granules viz. primary, secondary and tertiary granules. The primary granules or alpha granules of the platelets contain many factors which are released once the clot is formed after injury. These granules begin degranulating within ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
During bone grafting using autogenous bone graft in a maxillary/mandibular continuity defect correction surgery, a sinus augmentation surgery, or a dental implant surgery; a cancellous cellular marrow graft is placed in a dead space filled with clotted blood. This dead space is hypoxic (PO2 of 5 to 10 mm Hg), acidotic (pH 4 to 6), and contains platelets, leukocytes, red blood cells, and fibrin in a complex network around the transferred osteocytes, endosteal osteoblasts, and marrow stem cells . There are very few cells for regeneration in this area. The tissue surrounding the operated area is normoxic (PO2> of 45 to 55 mm Hg) at physiologic pH (pH 7.42) and contains a population of structural cells, healing-capable stem cells (also in very small numbers), and cut capillaries with clots and exposed endothelial cells.
The process of regeneration starts with the growth factors released from the platelets, including PDGF, TGF-β, and IGF. PDGF specifically acts as a mitogen for osteoblast, endothelial cell, and mesenchymal stem cell proliferation. TGF-β is not only a potent mitogen for un-differentiated mesenchymal cells, but also promotes angiogenesis and osteoblastic differentiation. IGF acts on the endosteal osteoblasts that line the trabeculae of grafted cancellous bone ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
By the third day postoperatively, capillaries can be seen to penetrate the graft and by 17 to 21 days, the capillary penetration of the graft is complete and the osteoprogenitor cells have vastly increased in number. As the wound matures, the expression of PDGF is downregulated and macrophage-derived growth and angiogenic factors take over (days 5 to 7). Most of the actions of macrophage-derived growth and angiogenic factors are similar to PDGF and IGF. By the 4th week, most of the ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Thus, from the above discussion, it is clear that platelets play a vital role during initial wound healing. The platelet concentrates provide a high concentration of growth factors in the healing area, thus promoting the wound healing and regeneration.
Clinical applications
Bone regeneration:
1. Sinus lift grafting.
2. Ridge augmentation.
3. Repair of bone defects created by removal of teeth or small cyst.
4. Ridge preservation techniques.
5. Periodontal defects.
6. Closure of cleft lip and palate defects.
7. Repair of oro-antral fistulas.
8. Craniofacial reconstruction.
Soft-tissue regeneration:
1. Periosteal and connective tissue flaps.
2. Free connective tissue and gingival grafts.
3. Root coverage procedures.
4. Controlling soft tissue healing and tissue maturity.
Contraindications
1. Thrombocytopenia < 100,000 / mm3.
2. Diagnosed and treated anemia Hb < 10.0 g%.
3. Patients who have metastatic disease.
4. Presence of tumor in the wound bed.
5. History of platelet dysfunction.
6. Active wound infection and sepsis requiring systemic antibiotics.
7. Patients with poor prognosis associated with other disease process.
8. Patient with bovine sensitivity.
9. Patients with religious beliefs that prevent use of blood.
10. Procedures where other techniques may provide better results.
How it started?
Initially, platelet concentrates were used for the treatment and prevention of hemorrhage due to severe thrombocytopenia, which is often caused by medullar aplasia, acute leukemia or significant blood loss during long-lasting surgery. This platelet-rich infusion contains around 0.5 x 1011 platelets per unit and has been designated as platelet-rich plasma (PRP). Another use of blood-derived products was to seal wounds and stimulate healing. Fibrin glues, which contain concentrated fibrinogen (polymerization induced by thrombin and calcium), have been classically used for this purpose. The term “platelet-rich plasma (PRP)” was initially used in 1954 by Kingsley 11 to ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Knighton et al. in 1986 12, developed an efficient clinical application of platelet concentrates for the treatment of chronic non-healing cutaneous ulcers, with a preparation using a 2-step centrifugation procedure and named it as “platelet-derived wound healing factors” (PDWHF). In other articles published during the same time, the same technique was named “platelet-derived wound healing formula (PDWHF)”. Whitman et al. (1997) 13 in 1997 showed that platelet concentrates improved healing and can be used in place of fibrin glues. Since then there has been a lot of research on platelet concentrates. Marx et al. in 1998 14 used ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Dohan et al. (2006) 15, developed a technique to obtain a second family of materials initially called Platelet Rich Fibrin (PRF). In this technique, no anticoagulant is added to the blood. The blood is immediately centrifuged with moderate forces for 12 minutes. Three layers, then appear in the tubes: the red blood cells are gathered at the bottom, acellular plasma is at the top of the tube and a strongly polymerized fibrin clot called PRF is formed in between 16. The historic evolution of platelet concentrates has been summarized in the following table,
Evolution of platelet concentrates.
Researcher & year Contribution
Kingsley (1954) First used the term platelet-rich plasma (PRP).
Matras (1970) Introduced 'fibrin glue'. This glue by polymerizing fibrinogen with thrombin and calcium.
Knighton et al. (1986) Demonstrated that platelet concentrates successfully promote healing and they termed it as platelet-
derived wound healing factors (PDWHF).
Whitman et al. (1997) Obtained platelet concentrates and named it as “platelet gel”.
Marx et al. (1998) Used platelet-rich plasma for the reconstruction of maxillofacial bone and popularized their use in reconstructive procedure in the orofacial region.
Year 1999 A new type of plasma concentrates plasma rich in growth factors (PRGF) was introduced with the name Endoret (Victoria, Biotechnology Institute BTI, Spain). However, because of the lack of specific pipetting steps and also lack of ergonomics, there were significant issues with this technique
Choukroun et al. (2000) Developed another form of platelet concentrates without adding anticoagulant and named it as 'Platelet-rich fibrin' (PRF). It was stamped as a “second-generation” platelet concentrate.
Sacco (2006) Introduced a new concept of CGF (concentrated growth factors).
Everts (2006, 2008) Focused on the leukocyte component of platelet concentrates and described two forms non-activated and activated. The inactivated/non-activated product was called “platelet-leukocyte rich plasma (P-LRP) and activated gel was labeled platelet-leukocyte-gel” (PLG).
Dohan et al. (2009) Proposed first classification of platelet concentrates.
Sohn (2010) Introduced the concept of sticky bone.
Mishra et al. (2012) Proposed a new classification of platelet concentrated, which limited its application to sports medicine.
Tunalı et al. (2013) Introduced a new product called T-PRF (Titanium prepared PRF).
Choukroun (2014) Introduced an advanced PRF called A-PRF (claimed to contain more monocytes).
Mourão et al. (2015) Described the preparation of an injectable form of platelet concentrate, I-PRF.
To understand the types of platelet concentrates, we must know how these are obtained and processed.
Basic clinical procedure to obtain platelet concentrates
Platelet concentrates are obtained from autologous blood, subjecting it to gradient density centrifugation. The processing of PRP involves the sequestration and concentration of platelets, and, therefore many growth factors they contain. The strategy is to amplify and accelerate the effect of growth factors contained in platelets, which are the universal initiators of almost all wound healing processes 17. In all available PRP techniques, the blood is collected with or without anticoagulant, just before or during surgery and is immediately processed by ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
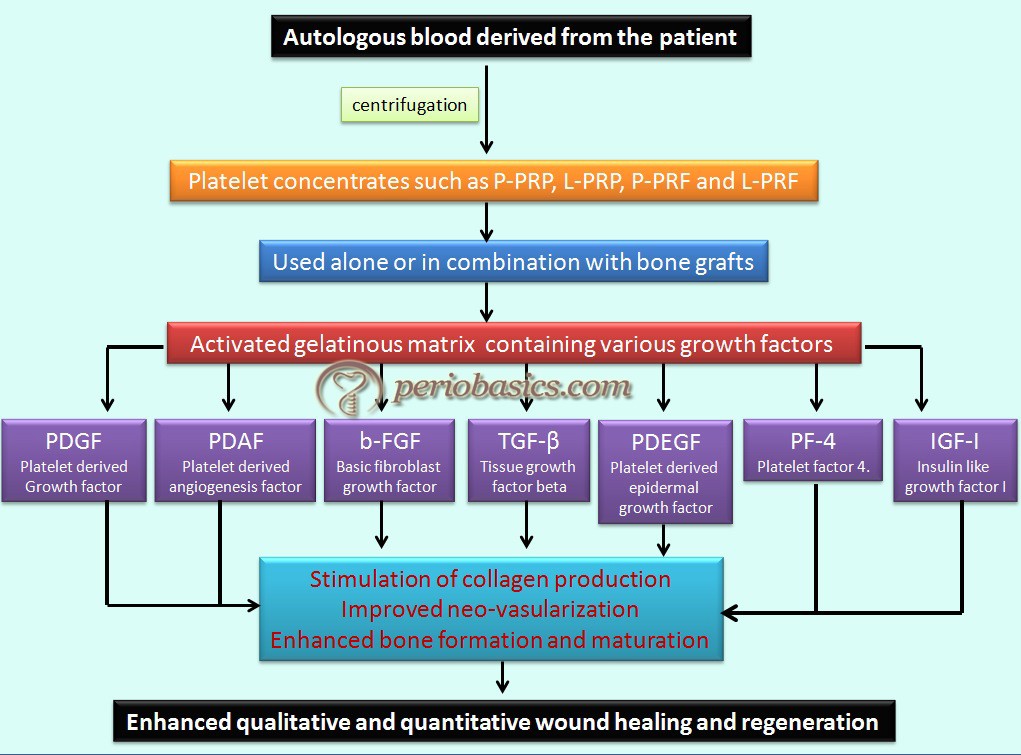
The first spin, i.e. “Hard spin” separates the PPP from the red fraction and platelet-rich plasma (PRP). The red blood cells (RBCs) are found at the bottom, acellular plasma (PPP, platelet-poor plasma) is in the supernatant and a ‘buffy coat’ of platelet-rich plasma appears in between. The second spin, i.e. “Soft spin” varies in different techniques, but is aimed at discarding both the RBC layer and the PPP to collect only the ‘buffy coat’ layer. Immediately prior to application, a platelet activator/agonist (topical bovine thrombin and 10% calcium chloride) is added to activate the clotting cascade, producing a platelet gel which is then applied to the surgical site. It must be noted that in many systems, the “Soft spin” is performed first followed by “Hard spin”.
In the recent past, many companies have come up with automated kits to obtain different types of plasma concentrates, exploiting their widespread ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Advantages of PRP
- It is a safe procedure. It is an autologous blood product, with no risk of infectious disease transmission or clerical errors, thus making it a safe product.
- It is non-invasive (other than phlebotomy) and painless procedure. No requirement for general anesthesia, and outpatient (dental office/treatment room) performance.
- No time-consuming visits to the blood bank for predonation. The sequester is done in the immediate preoperative period and utilized perioperatively.
- Accelerate endothelial, epithelial, and epidermal regeneration.
- Stimulate angiogenesis.
- Enhance collagen synthesis.
- Promotes enhanced soft tissue wound healing.
- Decreased dermal scarring.
- Provides for an immediate surgical hemostatic agent that is biocompatible, effective and safe with the enhanced hemostatic response.
Reverse the inhibition of wound healing caused by glucocorticoids. - High leukocyte concentration, adding an antimicrobial effect.
- The native fibrinogen concentration imparts a gelatinous adhesive consistency, for ease of surgical application.
- When mixed with crushed coral or crushed bone fragments, it forms a putty ideal for packing or structural reconstructions (as in mandibular reconstructions, maxillofacial procedures, dental implants) and actually improves handling characteristics of bone grafts.
- An augmented rate of extracellular matrix deposition, resulting in earlier wound closure.
Classification of platelet concentrates
Dohan et al. (2009) 19 have described the classification of platelet concentrates according to POSEIDO recommendations. According to the recommendations, irrespective of whatever be their form or cell content, all the products of this category are regrouped under the general term of “platelet concentrates”. Secondly, it is important to highlight the key influence of the leukocyte content 20-22 and fibrin architecture 23, 24 in the potential clinical or experimental effects of these products, and that each product refers to a specific biological imprint 25, 26. So, according to the recommendations, the platelet concentrates can be classified as follows,
1. Pure platelet rich plasma (P-PRP).
2. Leukocyte and platelet rich plasma (L-PRP).
3. Pure platelet rich fibrin (P-PRF).
4. Leukocyte and platelet rich fibrin (L-PRF).
Pure PRP (P-PRP)
To obtain pure PRP (P-PRP), PPP and superficial blood cells are transferred to another tube. After hard spin centrifugation, most of the PPP layer is discarded. The final P-PRP concentrate consists of an undetermined fraction of blood cell (containing a large number of platelets) suspended in some fibrin-rich plasma. Most leukocytes are not collected.
The method of collection of pure platelet-rich plasma was initially referred to as ‘plasmapheresis’, which used a cell separator. The machine utilizes an optical reader to detect the elements in serum. As soon as the integrated optical reader detects the first buffy elements in the serum, these are automatically collected into a separate bag as the platelet concentrate (PRP). This method allows around 40 ml of PRP to be obtained from 450 ml of whole blood. Blood is drawn into a collection bag containing a citrate-phosphate-dextrose anticoagulant. It is first centrifuged at 5,600 rpm to ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
PRGF/Endoret (Plasma Rich in growth factors, BTI BioTechnology Institute, Vitoria, Spain):
The scientific basis for the development of this system is that, upon activation with calcium chloride or autologous thrombin, platelets pour out their growth factor content to the local milieu. In addition, the fibrinogen present in plasma is cleaved to form fibrin and then crosslinked with factor XIIIa, creating a three-dimensional fibrin scaffold that retains a part of the released protein content, maintains the regenerative space and serves as a matrix for endogenous cells 27.
In this system, blood is collected into 9 ml tubes containing sodium citrate as an anticoagulant. Blood is then subjected to centrifugation at 1850 rpm for 8 minutes. The PPP is drawn off, avoiding the buffy coat. The remaining plasma is then activated with ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Leukocyte and platelet-rich plasma (L-PRP)
Leukocyte and platelet-rich plasma (L-PRP) contains high concentration of platelets, leukocytes, and other bioactive molecules, which play a prominent role in both bone and soft tissue healing processes. To obtain leukocyte-rich PRP (L-PRP), the PPP, the entire blood cell layer and some residual RBCs are transferred to another tube. After hard spin centrifugation, the PPP is discarded. The final L-PRP consists of most of the platelets and leukocytes, and residual RBCs suspended in some fibrin-rich plasma.
There are many kits available to obtain leukocyte and platelet-rich plasma (L-PRP) from autologous blood. Here is a brief description of some of them,
SmartPReP PRP (Harvest Corp, Plymouth, MA, USA):
It is a multifunction system with a specific collection and separation kit requiring little manipulation when used. This two-chamber device is designed to transfer automatically the upper layers (PPP and buffy coat) into the second chamber based on variations in weight and centrifugation speed. The centrifuge can also be used to concentrate stem cells from bone marrow aspirates 19. The final ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Curasan PRP kit (Curasan, Pharma GmbH AG, Lindigstrab, Germany):
In this system, 8 ml autologous whole blood is collected in the sample tube which is spun in a standard centrifuge for 10 minutes at 2400 rpm. This first spin separates the red blood cells from plasma. The PPP and the buffy coat of platelet-rich plasma are then transferred to a separate tube, which is then centrifugated for 15 minutes at 3600 rpm. 0.6-0.7 ml of pure PRP can be obtained from 8 ml of blood.
Friadent PRP (Friadent-Schütze, Vienna, Austria):
8.5 ml of blood is taken from the antecubital vein and is citrated to stop coagulation. The tube is then centrifuged for 10 minutes at 2400 rpm. Subsequently, the yellow plasma (containing the platelets) is taken up into a monovette with a long cannula using an additional air-intake cannula. It is then centrifuged for 15 minutes at 3600 rpm. According to the manufacturer, a high platelet concentrate with leukocytes can be obtained with this system.
Platelet-rich fibrin (PRF)
The platelet-rich fibrin was first developed in France by Choukroun et al. (2000) 28 for use in the field of oral and maxillofacial surgery. It is a second-generation platelet concentrate which contains platelets and growth factors in the form of fibrin membranes prepared from the patient’s own blood, free of any anticoagulant or other artificial biochemical modifications. It is one of the simplest and inexpensive methods of obtaining autologous platelet concentrate [centrifugation protocol (2700 rpm, 12 min)]. PRF has a dense fibrin network containing ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
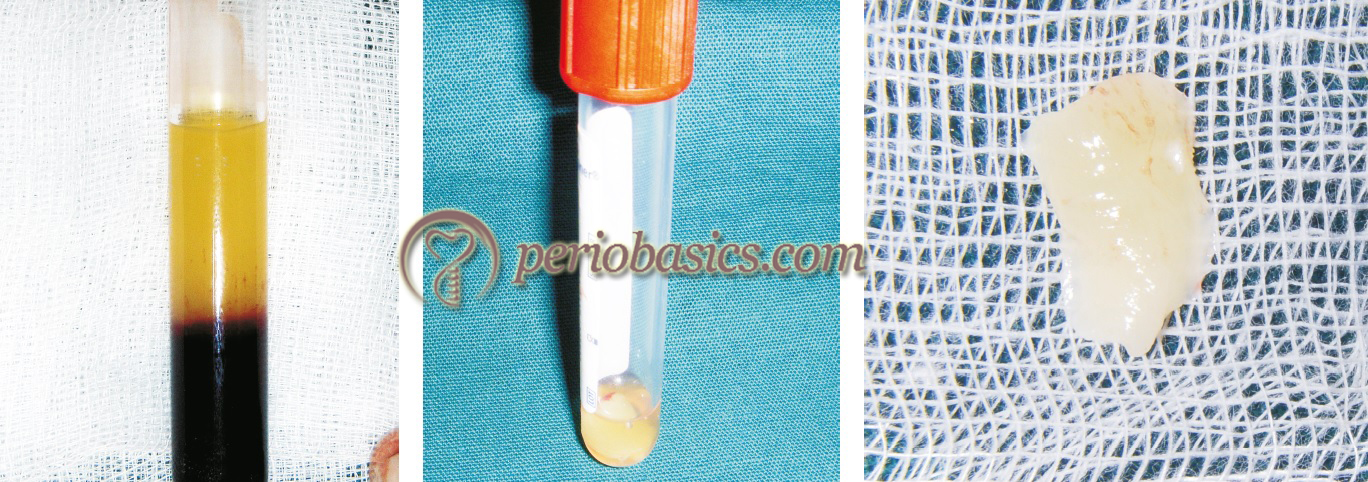
Advantages of PRF over PRP:
- No biochemical handling of the blood.
- Simplified and cost-effective process.
- Use of bovine thrombin and anticoagulants is not required.
- Favorable healing due to slow polymerization.
- More efficient cell migration and proliferation.
- PRF has a supportive effect on the immune system.
- PRF helps in hemostasis.
There are various commercially available systems to obtain PRF including,
Vivostat PRF (pure platelet-rich plasma).
Fibrinet PRF (without leukocytes).
Vivostat PRF (Vivolution, Alleroed, Denmark):
This is a fully automated system designed to obtain PRF from autologous blood perioperatively. This system comprises of following components:
PRF preparation unit: The PRF preparation unit is a device that extracts platelets and fibrin from the blood in the form of platelet-rich fibrin. A tubing assembly is attached to the PRF preparation unit.
Citrate/TA (PRF): A vial containing a sterile solution of citrate (anticoagulant) and 100 mg tranexamic acid (anti-fibrinolytic).
Codan spike: The dispensing spike is us to access the citrate vial and connect to the PRF preparation Unit.
Sugi swap: The two non-sterile Sugi swaps are used to clean the valve after the tubing assembly has been removed from the PRF preparation unit.
pH4: The pH4 syringe is loaded into the PRF preparation unit prior to processing. After processing, this syringe contains the PRF solution and is referred to as the fibrin syringe.
Replacement cap: The replacement cap is used to protect the luer tip of the fibrin syringe.
Disinfection swab: Used for disinfection of the rubber membrane on the citrate/TA (PRF).
Fibrinet PRFM kit (Cascade Medical, New Jersey, USA):
The kit contains two tubes, one for blood collection and another for PRFM clotting, together with a transfer device. The procedure involves the collection of 9 ml of venous blood from the antecubital vein in a collection tube which already contains trisodium citrate as an anticoagulant and a proprietary separator gel. The tube is then centrifuged for 6 minutes at high speed. It results in the formation of three distinct layers of RBCs, buffy coat, and PPP. The buffy coat and PPP are transferred to another tube containing CaCl2 and immediately centrifuged for 15 minutes. At the end of the procedure, a stable ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Leukocyte and platelet-rich fibrin (L-PRF)
The easiest and most inexpensive method to obtain leukocyte and platelet-rich fibrin (L-PRF) is Choukroun’s method. The Choukroun’s PRF is prepared by collecting 9 ml of blood from the patient immediately prior to the surgery. It is poured into a 10 ml tube without anticoagulant or gel separator, and is immediately centrifuged according to the following program: 30 sec acceleration, 2 min at 2,700 rpm, 4 min at 2,400 rpm, 3 min at 3,000 rpm, and 36 sec deceleration and stopping 28. When the blood comes in contact with the test tube wall the platelet gets activated, leading to the initiation of the coagulation cascade. After 10 min of centrifugation, 3 layers are obtained. The upper layer consists of acellular PPP, middle layer contains PRF clot and RBCs are at the bottom of the test tube. The fibrin clot obtained after centrifugation is removed from the tube and the attached red blood cells are scraped off from it and discarded.
The Intra-Spin L-PRF (Intra-Lock, Boca Raton, FL, USA) system has a three-step protocol for drawing and centrifuging the patient’s blood, removing the fibrin clot and processing it in the Xpression™ Fabrication Kit. According to the manufacturer, the system is capable of producing high-quality L-PRF matrix from autologous blood.
Literature review
Effect of platelet concentrates on wound healing:
It has been also proposed that PRP accelerates wound maturity and epithelialization, hence decreased scar formation. PDGF and epidermal growth factor (EGF) are the main growth factors involved in fibroblast migration, proliferation, and collagen synthesis. Increased concentrations of these growth factors are likely to be the reason for the accelerated soft tissue wound healing, which is suggested to be at least 2-3 times faster than that of normal 31.
Platelets in PRP also play a role in the host defense mechanism at the wound site by producing signaling proteins that attract macrophages 32. It has also been demonstrated that PRP may suppress cytokine release and limit inflammation, interacting with macrophages to improve tissue healing and regeneration 33 and promote new capillary growth 34.
Role of platelet concentrates in periodontal regeneration
Initially, it was shown by Choukroun et al. (2000) 28 that PRF used in implant surgery was able to enhance the healing properties of the bone. Chang et al. (2010) 35 elaborated the mechanism by which ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
A study done on human dental pulp cells demonstrated that PRF stimulated the osteogenic differentiation of these cells by upregulating osteoprotegerin and alkaline phosphatase expression 36. The PRF membrane has been used as a barrier membrane for guided tissue regeneration as a grafting material. It has been shown to have a good space making effect which facilitates cellular events that are favorable for periodontal regeneration, leading to the mineralized tissue formation. A randomized controlled trial done on 3-wall intrabony defects showed a statistically significant improvement in pocket depth reduction and bone fill in the test group (PRF) than in controls 37.
In a split-mouth study, 25 patients with intrabony periodontal defects were treated randomly using the surgical procedure with and without the application of autologous platelet concentrate. Radiological evaluation at 3 and 6 months postoperatively suggested a better bone density gain in the sites treated with the platelet concentrate. However, at 12-month post-surgery, bone density gain in the control sites appeared to be greater. The authors concluded that the improved periodontal regeneration in the test group as compared to controls was questionable 38.
Regenerative potential of platelet concentrates used with autogenous bone grafts
Marx et al. (1998) 14 reported the first clinical dental results of PRP in 1998. In this study, they placed 44 continuity bone grafts in the mandible without PRP and assessed them against 44 grafts placed with PRP at 2-month, 4-month, and 6-month maturity intervals with panoramic plain-film radiographs. They assessed platelet sequestration ability of the platelet concentrate process (gradient density centrifugation) and quantified the concentration as 338% of baseline platelet counts. Using a graft maturity index, each investigator assessed the radiographic graft maturation rate with PRP against its actual maturation rate without PRP. Investigators reported PRP grafts to be 2.16 times more mature at 2 months, 1.88 times more mature at 4 months, and 1.62 times more mature at 6 months. These differences were statistically significant (P = .001).
Anitua (1999) 39 demonstrated that extraction sites treated with a combination of autogenous bone and PRP demonstrated a better epithelialization and more compact mature bone with well-organized trabeculae than did the group receiving autogenous bone alone. In a randomized, blinded, prospective pilot study, four equal 8 mm diameter cranial bone defects were created and immediately grafted with autogenous bone, PRP alone, autogenous bone and PRP, and no treatment as a control. The defects were evaluated by digital subtraction radiography with step-wedge ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Also, many studies have reported contradictory findings questioning the effectiveness of PRP 41-43. Differences in the techniques of platelet concentrate preparation are one of the reasons which can alter their regenerative potential.
Regenerative potential of platelet concentrates used with allografts/xenografts/alloplasts:
Researchers have suggested that the addition of PRP to osteoconductive grafting materials can potentiate osteoinduction 44, 45. In one study, Aghaloo et al. (2002) 40 created 4 rabbit cranial defects in each of the 15 rabbits. They were grafted with either Bio-Oss, Bio-Oss with PRP, or autogenous bone or were left empty as a control. The histomorphometric evaluation showed that the addition of PRP significantly increased the percentage of bone ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
In a controlled clinical trial Jaske et al. (2003) 46, compared the combination of the xenograft bovine porous bone mineral (BPBM) and PRP versus BPBM alone and PRP alone. Six-month postoperative evaluations revealed superior regenerative effects in the PRP-treated groups.
Choukroun et al. (2006) 47 evaluated the potential of PRF in combination with freeze-dried bone allograft (FDBA) (Phoenix; TBF, France) to enhance bone regeneration in sinus floor elevation procedure. In this study, nine sinus floor augmentations were performed where, in 6 sites PRF was added to FDBA particles (test group), and in 3 sites FDBA without PRF was used (control group). Four months later for the test group and 8 months later for the control group, bone specimens were harvested from the augmented region during the implant insertion procedure. After 4 months of healing time, histologic maturation of the test group appears to be identical to that of the control group which was for a period of 8 months. Also, it was seen that ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
On the contrary, some studies have doubted the effectiveness of platelet concentrate used with bone grafts in regeneration. Aghaloo et al. (2005) 48 in one study compares bone healing in four cranial defects in the rabbit grafted with freeze-dried mineralized bone (FMB) alone, FMB+PRP, freeze-dried demineralized bone (FDDB) alone, and FDDB+PRP. In this randomized, blind, prospective pilot study, fifteen New Zealand white rabbits were utilized. Radiographic and histomorphometric analysis were done at 1, 2, and 4 months. This study failed to show a radiographic or histomorphometric increase in bone formation with the addition of PRP to either FMB or FDDB in non-critical-sized defects in the rabbit cranium.
Recent advances in platelet concentrates
Since their introduction in the early 1950s, platelet concentrates have been extensively used in the medical as well as dental fields. There have been many advances in obtaining the platelet concentrates in recent years. Table 72.2 has summarizes the journey of platelet concentrates. In the following discussion, we shall read about the recent advances in the platelet concentrates.
Concentrated Growth Factors (CGFs):
The concept of CGFs was introduced by Sacco in 2006 50. To prepare CGFs, a special centrifugation machine, Medifuge (Italy) is used. The process is similar to that used to obtain PRP but with a different centrifugation speed. This newer technique allows the separation of a fibrin matrix which is much denser, larger and richer in growth factors. The preparation protocol for CGFs consists of the following sequence,
30 seconds acceleration,
2 minutes at 2700 rpm/735 g,
4 minutes at 2400 rpm/580 g
4 minutes at 2700 rpm/735 g
3 minutes at 3000 rpm/905 g
36 seconds deceleration and stop.
Many studies have been done to evaluate the regenerative potential of CGF. In a study, Sohn et al. (2011) 58 evaluated the predictability of new bone formation in the maxillary sinus using autologous fibrin-rich blocks with concentrated growth factors (CGFs) alone as an alternative to the graft material. The authors concluded that fibrin-rich blocks with CGFs act as an alternative to bone grafting and can be a predictable procedure for sinus augmentation. In another study, the effect of CGF on implant stability and osseointegration was assessed by Pirpir et al. (2017) 59. It was observed in the study that ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Titanium-prepared platelet-rich fibrin (T-PRF):
Tunalı et al. (2013) 55, introduced a new product called T-PRF. During the conventional PRP preparation procedure, some researchers have pointed out the possible health hazards caused by silica particles which are small enough for a fraction to remain suspended colloidally in the buffy coat, fibrin, and platelet-poor layers of plasma 61. These particles might get an entry in the patient’s body when the product is used for treatment. T-PRF is a newer method of preparation of platelet concentrates which is based on the hypothesis that titanium tubes may be more effective at activating platelets than the glass tubes used in Chouckroun’s method 15, 19, 28, 62.
The centrifugation protocol is similar to that followed to obtain L-PRF (2700 rpm, 12 min). The basic histological structure of L-PRF and T-PRF is mostly similar to some difference in fibrin structure. In T-PRF the fibrin structure is more ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Advanced PRF (A-PRF):
It is well known that monocytes play a vital role in angiogenesis and bone regeneration. They are a good source of vascular endothelial growth factor (VEGF) and bone morphogenetic proteins. A-PRF was introduced by Choukroun (2014) 56 in an attempt to incorporate the monocytes within PRF. As compared to the standard centrifugation protocol (2700 rpm, 12 min) for the preparation of standard PRF, the centrifugation protocol for obtaining A-PRF is changed (1500 rpm, 14 min) 64. It was observed that with the lower centrifugation protocol, the presence of macrophages was improved in the PRF. That is why the name ‘advanced PRF’ was used for the PRF derived from this new protocol. The microscopic observations of A-PRF demonstrated a ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
Advanced platelet-rich fibrin (A-PRF) +:
Working on the low-speed concept, newer forms of PRF have been described. In A-PRF+, the centrifugation protocol was changed to 1300 rpm for 8 minutes. In a study, it was observed that A-PRF+ demonstrated significantly higher total growth factor release compared with A-PRF and L-PRF. The authors correlated his finding with the presence of a higher number of leukocytes contained within the A-PRF+ scaffolds centrifuged using lower G-forces and centrifugation times. However, these are preliminary findings and more animal and clinical studies are required to further confirm the effects of these results in vivo 65.
Injectable-PRF (i-PRF):
The Injectable form of PRF is obtained by centrifugation of whole blood at 700 rpm for 3-4 minutes. When a particulate bone graft is added to i-PRF, the result is the formation of a well agglutinated red-colored “sticky bone”. i-PRF has been shown to release the growth factors for ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
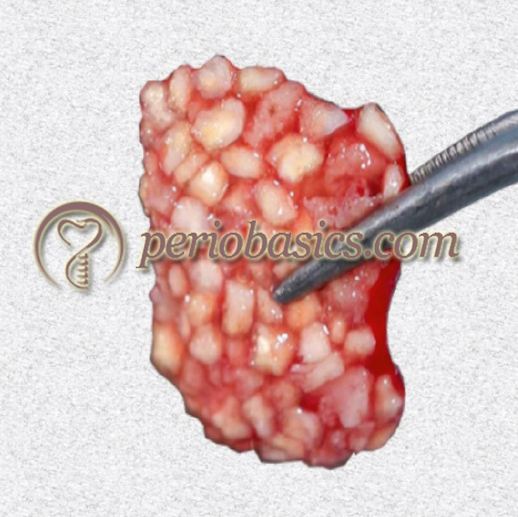
Advanced fibrin glue (AFG) and Sticky bone:
Fibrin glue is a two-component adhesive of fibrinogen and thrombin based on imitating the coagulation process of the body. In the last step of coagulation, thrombin facilitates the conversion of fibrinogen into fibrin, thereby forming a three-dimensional fibrin network. Fibrin glue is obtained by initiating the clotting process and is used to stop bleeding, seal wound edges with reduced scarring and for scaffolds in tissue engineering. Although commercial fibrin glue preparations are available, autologous fibrin glue offers advantages of reduced risk of contamination and immunological responses, as well as economic factors. To obtain autologous fibrin glue, 20- 60CC of blood in non-coated tubes is centrifuged at 2400-2700 rpm for 2 min. Out of the two layers obtained, the deeper layer is RBC’s and the superficial layer is AFG.
“Sticky bone” is based on the concept of fabricating growth factors-enriched bone graft matrix using autologous fibrin glue. The process of obtaining AFG has been stated above. The AFG thus obtained is then ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……….
As already stated, sticky bone can also be prepared by using i-PRF. The difference lies in the microstructure and the biological properties of the resultant product.
Conclusion
The application of platelet concentrates in periodontal regeneration has been extensively studied. As discussed above, many studies show significant improvement in periodontal regeneration after platelet concentrate application, whereas many others show no significant improvement. Since platelets play an important role during wound healing, platelet concentrates can be used to improve healing. Presently, dozens of kits are commercially available exploiting the widespread popularity of platelet concentrates. Irrespective of what the manufacturers claim, one need to go through the literature to judicially apply these concentrates for better clinical results.
References
References are available in the hard-copy of the website.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

