Introduction to relationship between cardiovascular and periodontal diseases
Periodontal infection is found worldwide and is among the most prevalent human microbial diseases. It has been mentioned that severe generalized periodontal disease is present in 8% to 13% of the world’s total adult population 1. The roles of infection and inflammation in atherosclerosis have become increasingly apparent. Over the last three decades, several studies have reported epidemiological associations between periodontitis and cardiovascular diseases 2-5. On the other hand, some studies have shown a weak or no association between periodontitis and cardiovascular diseases 6-8. However, at present, most of the research is in favor of direct or indirect association of periodontitis and cardiovascular diseases.
A survey that evaluated the association between periodontal and cardiovascular diseases in the third National Health and Nutrition Examination Survey or NHANES III, found that the odds of having a history of heart attack increased with the severity of periodontal disease. The highest severity of periodontal disease in the population was associated with an odds ratio (OR), of 3.8 [95 percent confidence interval (CI) 1.5 to 9.7] as compared to no periodontal disease, after adjusting for age, sex, race, poverty, smoking, diabetes, high blood pressure, body mass index and serum cholesterol levels. Thus, this cross-sectional study confirmed the association seen in other cross-sectional studies, as well as in case-control studies, and also showed a direct relationship between heart disease and increasing levels of periodontal disease 9.
The cardiovascular diseases have a complex etiology which includes a set of genetic and environmental factors. The genetic factors primarily include genetic markers for hypertension, diabetes, marked obesity, lipid metabolism, fibrinogen levels and platelet P1 polymorphism. The environmental risk factors include diet, physical inactivity, stress, cigarette smoking, socioeconomic status, chronic infections, use of non-steroid anti-inflammatory drugs and possible endothelial cell injury. Let us now discuss, various cardiovascular diseases and their relation with periodontitis.
Atherosclerosis and coronary heart diseases
Before discussing the relationship between periodontitis and coronary heart disease, let’s first try to understand how atheroma develops in an artery. The stages in the development of atherosclerosis involve a series of events that include the development of fatty streak, progression to complex plaque and plaque rupture 14.
What is Atherosclerosis?
Atherosclerosis is a multifaceted, progressive, inflammatory disease that affects mainly large and medium-sized arteries. It is characterized by the formation and build-up of atherosclerotic plaques that consist of a well-defined structure of lipids, necrotic cores, calcified regions, inflamed smooth muscle cells, endothelial cells, immune cells and foam cells; consequently, atherosclerosis is associated with cardiovascular diseases 15.
Atheroma formation:
It is now well documented that first step in the formation of an atheroma is most likely, the endothelial injury. After the endothelial injury, the surface adhesion molecules in the endothelial cells are up-regulated which lead to monocytes adhesion. The adherence of monocytes to endothelium is facilitated through the expression of MCP-1 (Monocyte chemoattractant protein-1). These monocytes recruited from the blood stream, then enter the vessel wall (intima layer) and become macrophages. These activated macrophages secrete various growth factors and cytokines, which lead to smooth muscle proliferation in the blood vessel wall. The cytokines secreted by activated macrophages further stimulate endothelial cells. These macrophages in the vessel wall accumulate low-density lipoproteins (LDL) in the oxidized or modified form. These LDL can undergo progressive oxidation. With the formation of lipid peroxidases and accumulation of cholesterol esters, the result is the formation of “Foam cells”. Modified LDL are chemoattractants for monocytes, which lead to further accumulation of these cells and thereby leading to the formation of foam cells. At this stage, this structure is called “Fatty streak”.
This fatty streak, now becomes a …….. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book……….
Now, if this fibrotic cap becomes thin and ruptures, there is activation of “clotting system” leading to thrombus formation which clinically precipitates as myocardial infarction.
Proposed pathways linking periodontal diseases to coronary heart disease (CHD)
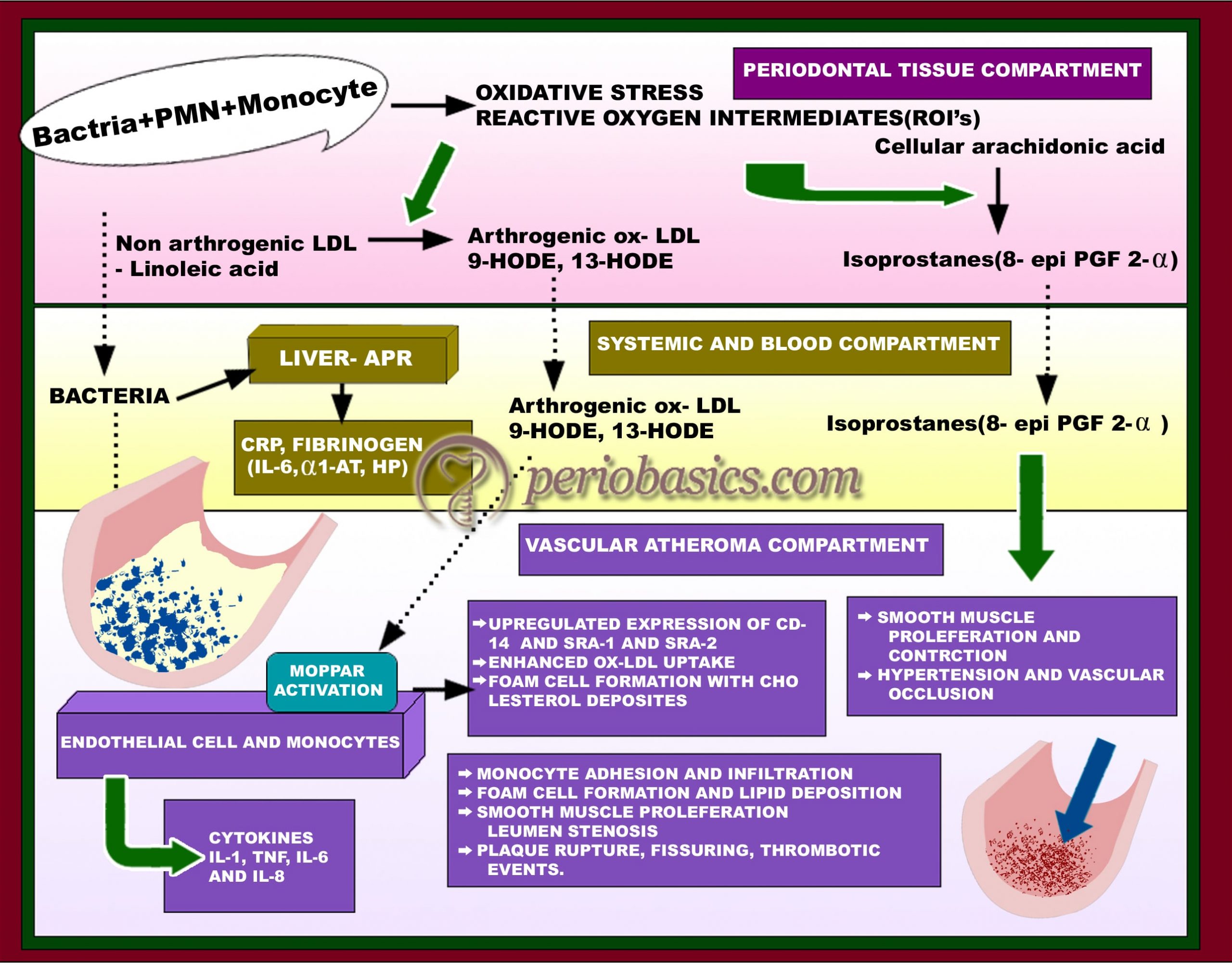
Periodontal diseases might affect heart disease through various mechanisms including direct involvement of oral bacteria, indirectly by bacterial toxins, and by inducing inflammatory mediators that enter the bloodstream and contributing to chronic, systemic vascular challenge 16, 17. Bacterial DNAs of periodontal pathogens such as Porphyromonas gingivalis, Tannerella forsythia, Treponema denticola, Aggregatibacter actinomycetemcomitans and Campylobacter rectus have been detected in stenotic coronary artery plaque samples 18. The DNA of these microorganisms has also been detected in aneurysm walls and aneurysmal thrombus tissues 19, 20. Another possibility is that the inflammation caused by periodontal disease …….. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………..
Following is the description of some well-accepted mechanisms of periodontitis-associated cardiovascular diseases,
Effect on platelets:
It has been found that bacteria may promote platelet aggregation directly or indirectly. Platelets participate in the immune response against infection. Oral bacteria may affect platelet function by three mechanisms. The first mechanism involves platelet activation by the immune system. The immune response against bacteria in the oral cavity may stimulate platelet aggregation. The second mechanism involves the activation of platelets by bacterial products and the third mechanism involves direct attachment of bacteria to platelets resulting in their activation 22.
The in vitro studies [reviewed in Kerrigan and Cox (2010) 23] have demonstrated that many Streptococci species, particularly belonging to viridans group, S. sanguinis (formerly known as Streptococcus sanguis), S. gordonii, S. mutans, and S. mitis may promote platelet aggregation. The platelet aggregation by bacteria is mediated by various surface proteins such as platelet aggregation associated protein (PAAP) of S. sanguinis 24, adhesins such as SspA and SspB in S. gordonii 22, serine-rich glycoproteins designated SrpA and GspB/Hsa in S. sanguinis 25 and S. gordonii 26, respectively, as well as glucosyltransferases 27. The PAAP are collagen-like cell surface antigen containing the sequence KPGEPGK. This sequence forms a …….. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………..
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Invasion and/or uptake of bacteria in endothelial cells and macrophages:
P. gingivalis (that is considered as one of the important putative periodontal pathogens) can invade aortic and heart endothelial cells via its fimbriae 32. P. intermedia 33 and S. mutans 34 have also been shown to invade and persist within the aortic endothelial cells, in vitro. The invasive strains of P. gingivalis have been shown to induce their uptake by macrophages and enhance foam cell formation, in the presence of LDL, in vitro 35. The ability of these bacteria to survive in the intracellular environment may allow the establishment of secondary chronic infection, which may contribute to the pathogenesis of atherosclerosis. The invasion of endothelial cells by P. gingivalis has been shown to alter the nature of these cells.
The invaded endothelial cells demonstrate increased procoagulant properties, increased mononuclear cell adhesion, increased expression of cell adhesion molecules, and increased production of pro-inflammatory cytokines and chemokines such as, IL-6, IL-8, and MCP-1 36, 37. Furthermore, the invasion of endothelial cells by P. gingivalis has been shown to induce apoptotic cell death of these cells 38. Thus, invasion of endothelial cells by oral bacteria induces cellular dysfunction, which may be associated with the pathogenesis of atherosclerosis. Macrophages, which have an important function in …….. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………..
Effects of pro-inflammatory mediators:
It has been well established that during host-microbial interactions in periodontal diseases, various pro-inflammatory cytokines are released which may enter the systemic circulation and affect other organ systems. Furthermore, it has been found that inflammatory mediators such as, tumor necrosis factor (TNF)-α, interleukin (IL)-1β and prostaglandin E2 (PGE2) play a key role in human coronary heart disease and atherosclerosis 39. These cytokines promote cholesterol accumulation in monocytes and smooth muscle proliferation, which presumably results in the thickening of vessel walls 40, 41. Epidemiological studies have shown that increased levels of cytokines (IL-1, IL-6, TNF-α, and MCP-1), and acute-phase proteins such as C-reactive protein (CRP) and fibrinogen 42-44 are associated with …….. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………..
Autoimmune response:
It has been shown that endotoxins of plaque microorganisms are capable of penetrating the gingival tissues and entering into the blood stream, in amounts sufficient to bring about a systemic LPS-specific antibody response 49. Heat shock proteins (HSP) are produced by a wide variety of bacteria and human cells under a variety of stressful or harsh conditions such as high temperature, infection, inflammation, and mechanical stress 50. The bacterial HSPs are also known as GroEL 51. These are highly conserved proteins and many periodontal pathogens express HSPs that are homologous to human HSPs 52. Due to their homogenous nature, the antibodies produced against periodontal bacteria may cross-react with human HSPs. GroEL proteins and homologues have been identified in several oral bacteria including P. gingivalis, A. actinomycetemcomitans, F. nucleatum, P. intermedia, T. denticola, and S. mutans [reviewed by Goulhen et al. (2003) 53]. It has been shown that antibodies developed against P. gingivalis HSP60, cross-react with human HSP because of the structural homology that exists between the two 54, 55. Another study demonstrated that GroEL from P. gingivalis, F. nucleatum, and A. actinomycetemcomitans was recognized by serum antibodies in patients with periodontal disease 56.
A positive association between elevated levels of antibodies to human HSP60 with the severity of coronary artery disease 57 and atherosclerosis 58 has been demonstrated. Furthermore, periodontal treatment has been shown to significantly reduce the levels of anti-P. gingivalis GroEL antibodies in patients with moderate to advanced periodontal disease, while the mean levels of antibodies to human HSP60 remained unchanged 59. These findings were supported by Buhlin et al. (2009) 60.
It should be noted that …….. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………..
Our current understanding of the association between periodontitis and coronary heart disease
It has been observed that bacteria entering the systemic circulation from periodontal sites may contribute to the development of atherosclerosis. The non-surgical periodontal therapy has been shown to cause transient bacteremia which causes initial, transient elevation in inflammatory and pro-thrombotic mediators and an overall decrease of endothelial function 61. Furthermore, following completion of the treatment, a reduction in the inflammatory markers, both, at …….. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………..
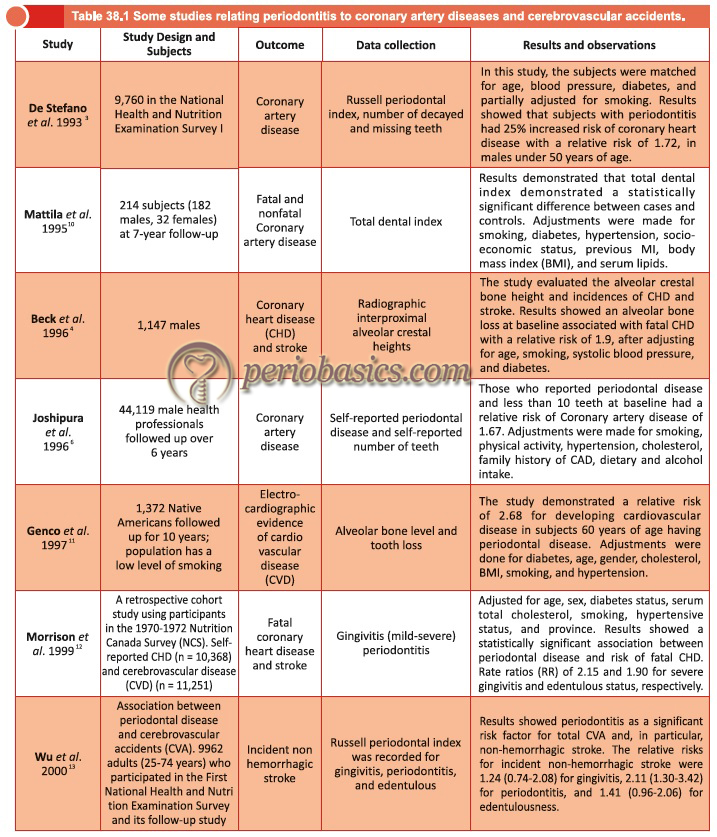
Although the mechanism by which microbial overload in periodontal tissue leads to cardiovascular vasculature involvement is quite complex, Offenbacher et al. (1999) 63 proposed a working model of periodontitis associated atherosclerosis. They explained the following mechanism of the association between periodontal microbial overload and atherosclerosis,
- Periodontal infection usually involves microorganisms like P. gingivalis, T. forsythia, A. actinomycetemcomitans and others.
- These microorganisms produce various virulence factors, because of which the host immune response is activated.
- It leads to the recruitment of inflammatory cells like, PMNs and macrophages in that area.
- Interactions between the host immune cells and invading microorganisms lead to the production of reactive oxygen intermediates, which cause increased oxidative stress.
- Now, these reactive oxygen intermediates which include oxidized low-density lipoproteins (ox-LDL) and products of arachidonic acid metabolism (Isoprostanes), reach the systemic circulation.
These products on reaching the site of atheroma, stimulate various functions like smooth muscle proliferation, upregulation of cellular receptors for monocyte recruitment, enhanced ox-LDL uptake and thus formation of more and more foam cells. As this process continues, there is stenosis of artery lumen and rupturing of plaque from arterial wall leads to a thrombotic event. Another working model of the potential mechanism of role of periodontal infectious agents in atherosclerosis has been proposed by Fong (2002) 64. The author explained the direct and indirect mechanisms of the potential role of periodontal bacteria in the development of atherosclerosis. The direct mechanism involves the ingress of bacteria and their products in the systemic circulation and their participation in the formation of atherosclerotic plaque. This mechanism is supported by the findings that …….. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………..
Infective endocarditis
Bacterial endocarditis is an infection of the inner surface of the heart or the heart valves, caused by bacteria, usually found in the mouth, intestinal tract or urinary tract. It can seriously damage the heart valves and cause other serious complications if it is not treated quickly with antibiotics.
Certain oral bacteria are known to be the causative agents associated with infective endocarditis 3. Oral bacteria Streptococcus viridans is responsible for about 50% of all bacterial endocarditis cases. Other common culprits include Staphylococcus aureus and Enterococcus. Staphylococcus aureus can infect normal heart valves and is the most common cause of infectious endocarditis in intravenous drug users. Other causative microorganisms for infective endocarditis include Enterococci, which are occasionally found in the oral cavity, or Gram-negative HACEK microorganisms (Haemophilus species, Actinobacillus actinomycetemcomitans, Cardiobacterium hominis, Eikenella, and Kingella), some of which, especially A. actinomycetemcomitans and Eikenella corrodens, are putative periodontal pathogens.
The bacteria implicated in bacterial endocarditis have been shown to originate from periodontal infections. During routine dental procedures like scaling and root planing, dental extraction, etc. these microorganisms can get entry into the systemic circulation and can populate the endocardium or the cardiac valves. Transient bacteremia is well documented following tooth extraction 71, and following treatments for periodontal diseases such as …….. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………..
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Valvular heart diseases
Prosthetic valve endocarditis (PVE) is an endovascular, microbial infection occurring on parts of a valve prosthesis or on reconstructed native heart valves 78. It is a serious complication of cardiac valve replacement, reported to occur in 1% to 9.4% of patients 79-83. Prosthesis made from metal, pyrolytic carbon or other materials do not allow adherence of microorganisms as long as they are free from the thrombotic material. Infections of mechanical prosthesis generally originate from the sewing cuff or from thrombi located near the sewing ring downstream in recirculation areas. Inflammatory periprosthetic leaks, ring abscesses, and invasion of the infective process into the adjacent tissue are common findings.
These patients should be given antibiotic prophylaxis before starting dental treatments which can lead to transient bacteremia. The microbiology of PVE is very different from that of native valve endocarditis (NVE). Streptococci and Enterococci occur less frequently, while Staphylococci, bacteria of the HACEK group (Haemophilus, Actinobacillus, Cardiobacterium, Eikinella, and Kingella), and fungi are found more frequently in cases of PVE.
Myocardial Infarction
As already discussed, the periodontal infections have direct and indirect effects on systemic conditions, directly by microbes and their products and indirectly by chemical mediators or pro-inflammatory cytokines. According to the most widely established hypothesis, the relationship between acute myocardial infarction and periodontitis depends on the risk factors, common to both the diseases 84, with tobacco use as the main confounding factor 85. Smoking …….. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………..
It has also been proposed that periodontal pathogens or their lipopolysaccharides are systemically disseminated via the blood flow and directly infect the vascular endothelium, producing an atherosclerotic lesion and subsequent myocardial ischemia 15, 91. The endotoxins produced by these microorganisms, along with inducing the production of pro-inflammatory cytokines also induce the production of acute-phase proteins in the liver, such as C-reactive protein (CRP). These proteins can form deposits in damaged blood vessels, with the consequent activation of phagocytes and release of nitrous oxide, contributing to the formation of atheromas 92.
Know More…
Myocardial infarction and C-Reactive protein:
CRP is a plasma protein involved in the acute phase response. CRP, named for its capacity to precipitate the somatic C-polysaccharide of Streptococcus pneumoniae, was the first acute-phase protein to be described and is an exquisitely sensitive systemic marker of inflammation and tissue damage 97. The specific ligand was later found to be phosphocholine. It is an important marker of disease activity. CRP has been found to interact with various other ligands, activate the classical complement pathway, stimulate phagocytosis and bind to FcγR immunoglobulin receptors.
Plasma CRP is produced only by hepatocytes, predominantly under transcriptional control by the cytokine IL-6. Although, other sites of local CRP synthesis and possibly secretion have also been suggested. The plasma half-life of CRP is about 19 hours and is constant under all conditions of health and disease.
Tissue necrosis is a potent acute-phase stimulus, and following myocardial infarction, there is a major CRP response, the magnitude of which reflects the extent of myocardial necrosis 98. Furthermore, the peak CRP values at around 48 hours after the onset, powerfully predict the outcome after myocardial infarction 99-101 and compelling experimental evidence now suggests that the CRP response not only reflects tissue damage in this context but may also contribute significantly to the severity of ischemic myocardial injury 102.
CRP has been shown to be not only a prognostic indicator of acute coronary syndromes, but also a predictor of future coronary events. Perhaps greater importance is the demonstration that CRP concentrations predict MI and stroke 93-95. So, it becomes important to identify those conditions that contribute to CRP level elevation that may have significant prognostic and treatment implications in patients with cardiovascular disease.
Studies done on microbial aspect, showed an association between periodontitis and acute myocardial infarction (AMI) in which periodontal destruction was correlated with the presence of periodontal pathogens. In particular, P. gingivalis might be considered as a potential risk indicator for AMI 96.
Conclusion
The epidemiological data has provided us with the evidence that periodontal diseases may act as risk factors for cardiovascular diseases. Furthermore, intervention studies have demonstrated that periodontal therapy can reduce the systemic markers of inflammation and may promote systemic health. However, the exact mechanism, how the periodontal infections can act as a risk factor for or contribute causally to cardiovascular diseases is still not clear. The recent consensus report on periodontitis and atherosclerotic cardiovascular diseases has recommended that patients with moderate to severe periodontitis should be informed that periodontal infections act as a risk factor for cardiovascular diseases and that the patients with more than one cardiovascular risk factors should undergo a medical evaluation of the risk factors 103. Because of extensive research going on in this field, more clarity in the mechanisms involved in the association of periodontal diseases to cardiovascular diseases is expected in future.
References
References are available in the hard-copy of the website.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

