Introduction to the complement system
Term ‘complement’ means to augment the effect of other components of the immune system. The complement system is a group of more than 23 proteins that interact with each other to opsonize the pathogen and induce a series of inflammatory responses that help to generate an immune response against that pathogen. Many of these proteins are proteases that are themselves activated by proteolytic cleavage. The principal participants in this system are 11 proteins designated C1 through C9, B, and D.
Historical aspect:
The complement system is an important component of the innate immune response. Buchner (1891) 21 found a heat-labile factor in blood that was capable of killing bacteria and named it ‘alexin’ (in Greek, means ‘to ward off’). In 1895 Bordet discovered that this heat-labile activity in serum was responsible for the lysis of bacteria. Vibrio cholerae were lysed within minutes when serum from an immunized animal was added to the bacteria. However, when this serum was incubated at 56°C for a few minutes, the lytic activity was lost even though the antibody activity remained. Untreated serum, when added to the reaction restored the lytic activity. Therefore, the lysis of bacteria required both the antibody and this heat-labile substance 22. Ehrlich (1899) named this heat-labile substance as ‘Complement’ 23.
Bordet described the antibody-dependent arm of the complement system, referred to as the ‘classical pathway’. An antibody-independent mechanism of the complement system was discovered by Pillemer in 1954 and he named it “properdin system” 24, 25. All components of the classical pathway and the membrane attack complex (MAC) are designated by the letter C followed by a number. The components were numbered in the order of their discovery rather than the sequence of reactions, which is C1, C4, ……… Contents available in the book…… Contents available in the book…… Contents available in the book…… Contents available in the book…..
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Activation of the complement system
Components of the complement systems are pro-enzymes. These components are cleaved to form active enzymes. Complement system can be activated by immune complexes and immunologic molecules like endotoxins. It occurs by 3 mechanisms,
1. Classical pathway.
2. Alternative pathway.
3. Lectin pathway.
Pathways of complement system activation
Classical pathway
The binding of the antibody to its antigen triggers the complement system through the so-called classical pathway. It can occur in a solution or when the antibodies have bound to antigens on a cell surface. Only IgM and IgG can activate the complement cascade. One molecule of IgM can activate the complement, but activation by IgG is with the help of the cross-linking of two molecules. C1 binds to the region located in the FC region of the heavy chain. Out of all IgG’s only IgG1, IgG2, and IgG3 can fix the complement and not IgG4.
Step by step activation of the classical pathway
C1 activation:
C1, binds to the Fc region of IgG or IgM antibody molecules that have interacted with the antigen. C1 binding does not occur to antibodies that have not been complexed with the antigen and binding requires the presence of calcium and magnesium ions (in some cases C1 can bind to aggregated immunoglobulin [e.g. aggregated IgG] or to certain pathogen surfaces in the absence of an antibody). The binding of C1 to the antibody is via C1q. C1q must cross-link at least two antibody molecules before it is firmly fixed. The binding of C1q results in the activation of C1r which in turn activates C1s. The result is the formation of an activated “C1qrs”, which is an enzyme that cleaves C4 into two fragments C4a and C4b.
C4 and C2 activation (generation of C3 convertase):
The C4b fragment binds to the membrane and the C4a fragment is released into the microenvironment. Activated “C1qrs” also cleaves C2 into C2a and C2b. C2a binds to the membrane in association with C4b, and C2b is released into the microenvironment. The resulting C4bC2a complex is a C3 convertase, which cleaves C3 into C3a and C3b.
C3 activation (generation of C5 convertase):
C3b binds to the membrane in association with C4b and C2a, and C3a is released into the microenvironment. The resulting C4bC2aC3b is a C5 convertase. The generation of C5 convertase is the end of the classical pathway. It results in the formation of C5a and C5b where C5a diffuses away and C5b participates in the formation of ‘membrane attack complex’ (MAC). MAC that is responsible for the lysis of the bacterial cell is formed by C5b, C6, C7, C8 and C9.
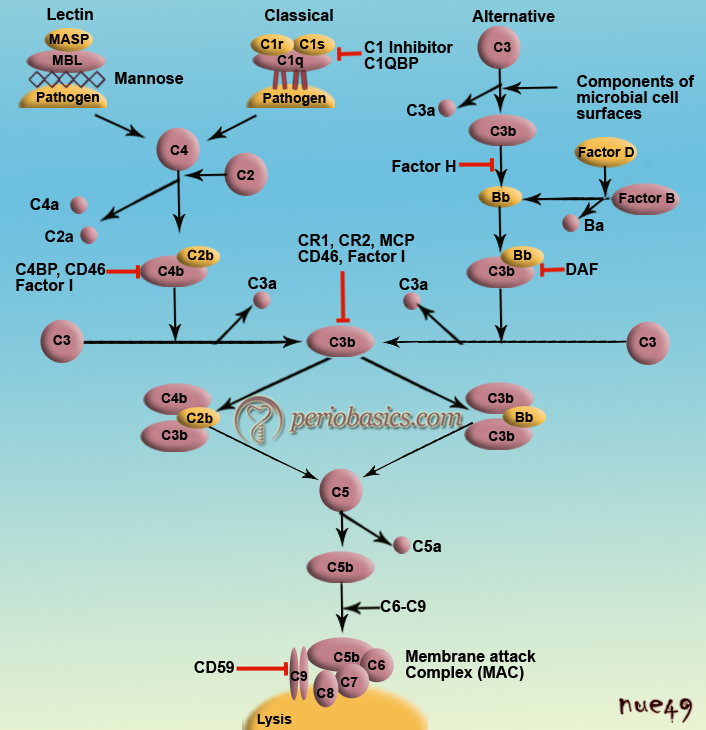
Alternative pathway
This pathway is activated in the presence of immunogenic bacterial, fungal and viral cell surface substances. There is no antibody present, as it is a new infection. The cell wall polysaccharides and endotoxins possess binding to C3 and factor B. This complex is cleaved by a protease, factor D to produce C3bBb. The C3bBb thus formed, acts as C3 convertase to form more C3b resulting in the formation of C3bBb3b which then acts as C5 convertase. It must be noted here that alternate pathway is more important when we first time get infected by microorganisms and the antibodies against them, required to trigger the classical pathway are not present.
Lectin binding pathway
The lectin binding pathway is very similar to the classical pathway. It is initiated by the binding of mannose-binding lectin (MBL) to the bacterial surfaces with mannose-containing polysaccharides (mannans). Binding of MBL to a pathogen results in the association of two serine proteases, MASP-1 and MASP-2 (MBL-associated serine proteases). MASP-1 and MASP-2 are similar to C1r and C1s, respectively, and MBL is similar to C1q. Formation of the MBL/ MASP-1/MASP-2 tri-molecular complex results in the activation of the MASPs and subsequent cleavage of C4 into C4a and C4b. The C4b fragment binds to the membrane and the C4a fragment is released into the microenvironment. Activated MASPs also cleave C2 into C2a and C2b. C2a binds to the membrane in association with C4b and C2b is released into the microenvironment. The resulting C4bC2a complex is a C3 convertase, which cleaves C3 into C3a and C3b. C3b binds to the membrane in association with C4b and C2a and C3a is released into the microenvironment. The resulting C4bC2aC3b is a C5 convertase. The generation of C5 convertase is the end of the lectin pathway. The biological activities and the regulatory proteins of the lectin pathway are the same as those of the classical pathway.
Regulation of the complement system
At antibody level:
Normally complement binding site located in the heavy chain on Fc region of antibodies, is not available for C1 component of the complement system. It is only after the binding of antigen, conformational changes take place which allows C1 complement to bind to the antibody. That is why the complement is not activated even when a high amount of IgM and IgG are present in the blood.
Regulation by serum proteins:
C1 Inhibitor (C1-INH) is an important regulator of the complement system. It inhibits the C1r and C1s serine proteases and prevents the auto-activation of the C1qrs complex. C1-INH is also a biologically significant inhibitor of kallikrein and coagulation factor XII. Its synthesis is stimulated by interferon-γ, interleukin 1 (IL-1) and IL-6.
C3a inactivator (C3a-INA; Carboxypeptidase B):
It inactivates C3a.
Factor H:
Its principal function is to regulate the alternative pathway of the complement system. It accelerates the decay of the alternative pathway C3 convertase (C3bBb) and is also a cofactor for factor I-mediated cleavage and inactivation of C3b. In the absence of Factor H, spontaneous activation of the alternative pathway of complement occurs in plasma, which leads to the consumption of complement components C3 and factor B.
Decay accelerating factor (DAF):
DAF is a glycol protein located on the surface of human cells and protects them from lysis by the MAC. It acts by destabilizing C3 convertase and C5 convertase.
C4 binding protein (C4-BP) and Factor I:
C4-BP facilitates the degradation of C4b by Factor I. C4-BP also prevents the association of C2a with C4b, thus blocking the formation of C3 convertase.
Protein S (vitronectin):
After the formation of MAC, some of the C5bC6C7 complexes formed can dissociate from the membrane and enter the fluid phase. They can bind to other nearby cells and lead to their lysis. This damage is prevented by Protein S (vitronectin). Protein S binds to the soluble C5b67 and prevents its binding to other cells.
| Component | Action | Control |
|---|---|---|
| C2a | Proinflammatory, accumulation of fluids causes edema. | C1-INH |
| C3a | Basophil and mast cells degranulation; enhanced vascular permeability, smooth muscle contraction. Anaphylatoxin. | C3a-INA |
| C3b | Opsonin, phagocyte activation. | Factors H and I |
| C4a | Enhanced vascular permeability, causes Basophil and mast cells degranulation and smooth muscle contraction. Acts as an Anaphylatoxin. | C3a-INA |
| C4b | Opsonin causes Phagocytosis | C4-BP and Factor I |
| C5a | It leads to Chemotaxis, stimulation of respiratory burst, activation of phagocytes and stimulation of inflammatory cytokines. Enhanced vascular permeability and causes Basophil and mast cells degranulation and smooth muscle contraction. Acts as an Anaphylatoxin. | C3a-INA |
| C5bC6C7 | Chemotaxis, Attaches to other membranes. Causes Inflammation and tissue damage. | Protein S (vitronectin) |
Biological effects of the complement components
Opsonization:
Phagocytosis of cells, antigen-antibody complexes, viruses, and other foreign invaders is much better in the presence of C3b. As C3b receptor is present on the surfaces of many macrophages, hence, C3b facilitates opsonization.
Chemotaxis:
C5a and C5b67 complex attracts neutrophils. They migrate well towards C5a which enhances the adhesiveness of the neutrophils to the endothelium.
Anaphylatoxins:
C4a, C3a, and C5a (in increasing order of activity) are all anaphylatoxins. They cause degranulation of the mast cells with the release of mediators e.g. histamine (increases vascular permeability, causes smooth muscle contraction, and broncho-spasm), but this anaphylaxis is less severe than anaphylaxis caused by IgE.
Cytolysis:
Insertion of C5b6789 complex into the cell membrane causes lysis of the cell. This process is not mediated by enzymatic degradation.
Enhancement of antibody production:
Binding of C3b with antigen on the surface of B-cells markedly enhances the production of antibodies, as compared to when only the antigen is attached to B-cell.
Disorders related to the complement system
- Inherited deficiency of some factors like C5-C8 greatly enhances susceptibility to infection by Neisseria.
- Inherited deficiency of C1 esterase inhibitors results in angioedema.
- Inherited deficiency of DAF results in paroxysmal nocturnal hemoglobinuria.
- If blood transfusion is mismatched, antigen-antibody reaction occurs that leads to the formation of membrane attack complex which causes hemolysis and anaphylatoxins cause shock
- Immune complex bind complement and thus complement levels are less in immune complex diseases for e.g. Acute glomerulonephritis and systematic Lupus erythrocytosis. Binding of complement to PMN’s causes the release of enzyme-tissue damage.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

