Introduction to Aggregatibacter actinomycetemcomitans
This bacterium was first isolated by Klinger 81 from a cervicofacial actinomycotic lesion in 1912 and was named Bacterium actinomycetemcomitans. Topley and Wilson (1929) 82 reclassified it as Actinobacillus actinomycetemcomitans and Potts et al. (1985) 83 as Haemophilus actinomycetemcomitans. In 2006 it was again reclassified based on multilocus sequence analysis by Nørskov-Lauritsen N and Kilian M (2006) 84. Because of the phylogenetic similarity of Actinobacillus actinomycetemcomitans, Haemophilus aphrophilus and Haemophilus segnis, genus Aggregatibacter (aggregate, to come together; bacteria, bacterial rod; aggregatibacter, rod-shaped bacterium that aggregates with others) was added to the family pasteurellaceae. This genus includes Gram-negative, non-motile, facultatively anaerobic rods or coccobacilli that were previously known as Haemophilus.
Morphological Structure
Structurally, A. actinomycetemcomitans are Gram-negative, small non-motile coccobacilli that grow singly, in pairs, or in small clumps and are described as fastidious and CO2-requiring bacteria. These are small, short (0.4×1 µm) straight or curved rods with rounded ends 85. Electron microscopic studies have demonstrated membrane vesicles that appear to be released from the cells. On blood agar, colonies are small, gray to white, translucent, smooth and non-hemolytic. Growth is stimulated by the addition of CO2 and species grows well in 5-10 % of CO2 86. These ferment a range of sugars, including glucose and fructose, but not sucrose and lactose. Acid end products include lactate, succinate, acetate and propionate. A. actinomycetemcomitans is grown in anaerobic cultures and from plaque, it can be rapidly identified by fluorescent labelled antisera. It can also be identified by the use of DNA probes.
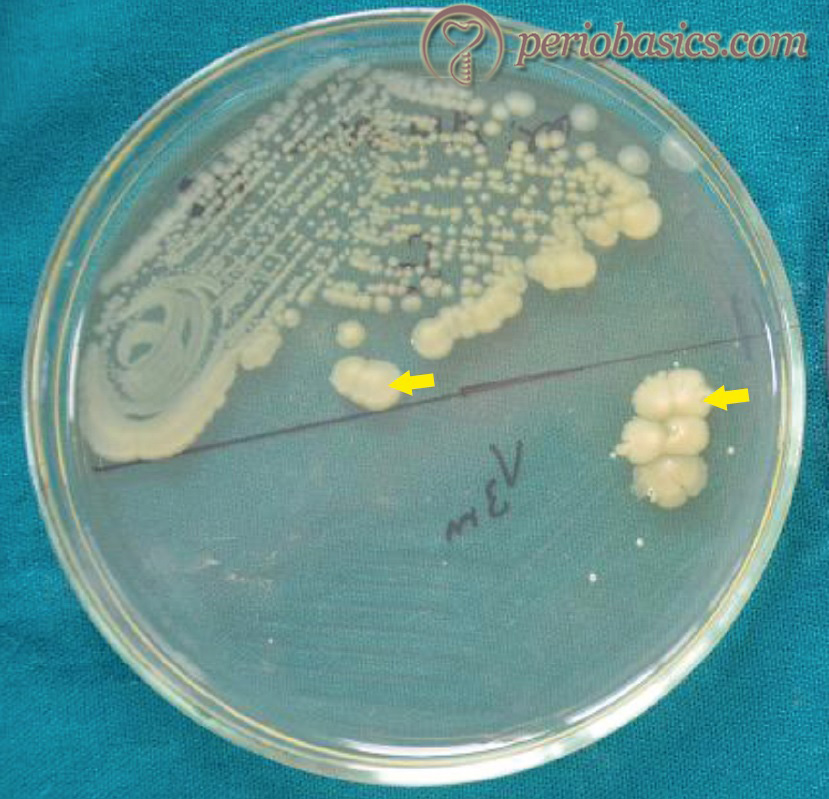
When grown in culture, these form small colonies, approximately 0.5-1.0 mm in diameter. Colonies are translucent (or transparent) with ……Contents available in the book ……..Contents available in the book…………..Contents available in the book…………..Contents available in the book……..
Taxonomy of A. actinomycetemcomitans
King and Tatum (1962) 87 provided the first detailed biochemical and serological description of A. actinomycetemcomitans. As already stated, these bacteria are closely related to Haemophilus aphrophilus. Differentiation is done by its ability to grow in the absence of haemin and factor V (Nicotinamide adenine dinucleotide). Serological studies have been attempted to determine the antigenic relationship between A. actinomycetemcomitans and Haemophilus aphrophilus. Using agglutination studies, 24 different serogroups and 6 major agglutination antigens of A. actinomycetemcomitans were identified. Currently, A. actinomycetemcomitans strains are classified into ……Contents available in the book……..Contents available in the book…………..Contents available in the book…………..Contents available in the book……..
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Most leukotoxic strains are of serotype b. It is important to note that serotype b of A. actinomycetemcomitans has been found more frequently and detected in higher numbers in active periodontitis lesions, whereas serotype a and c have a stronger association with periodontal health 100. Taxonomic classification of bacterial serotypes is based on the following biochemical characteristic features:
1. Fermentation of different carbohydrates.
2. Multivariate analysis of chemotaxonomic data.
3. Cellular fatty acid composition.
4. The composition of lipopolysaccharides.
5. Analysis of total proteins.
6. Multilocus enzyme electrophoresis.
7. Respiratory quinones.
Surface ultrastructure of A. actinomycetemcomitans
The surface ultrastructure of A. actinomycetemcomitans demonstrates the following components,
Fimbriae:
They may exhibit fimbriae which are around 2 µm in length and 5 nm in diameter. Fimbriated strains produce colonies with a star-shaped interior structure. These are required for attachment of the bacterium to host.
Vesicles:
These are a prominent feature of A. actinomycetemcomitans. These structures are lipopolysaccharides in nature and originate from and are continuous with the outer membrane. A. actinomycetemcomitans species with high leukotoxic production have abundant extra-cellular membrane vesicles. Vesicles per se exhibit leukotoxic activity. Other components of vesicles are endotoxin, bone resorption activity, bacteriocin, and actinobacillin.
Extracellular amorphous material:
Certain A. actinomycetemcomitans have an amorphous material that frequently embeds adjacent cells in the matrix.
Virulence factors associated with A. actinomycetemcomitans
A. actinomycetemcomitans has many virulence factors that facilitate attachment and proliferation of these bacteria in the host. These include,
- Adhesion factors
- Bacteriocin
- Bone resorption factors
- Collagenases
- Cytotoxins
- Extracellular membrane vesicles
- Fc binding proteins
- Leukotoxin
- Lipopolysaccharides (LPS)
- Immunosuppressive factors
- Products that inhibit PMN function.
- Factors facilitating penetration of epithelial cells
- Cytolethal distending toxin
These are discussed as follows,
Adhesion of A. actinomycetemcomitans
It is executed by adhesins which are bacterial cell surface components. They interact and bind to very specific receptors in saliva, on the surface of the tooth, on extracellular matrix proteins and on epithelial cells. Distinct adhesins may be expressed by bacterium under different environmental conditions 102. Most of the adhesins are proteinaceous in nature. A. actinomycetemcomitans strains with fimbriae adhere three to four folds better than those without fimbriae.
- Bacterial aggregation during plaque formation: The bacterial aggregation during plaque formation is very specific. Bacteria assert in the plaque biofilm by means of aggregation, inter-generic and intra-generic coaggregation and interactions with distinct and bacteria-specific salivary binding proteins. Following initial colonization, bacteria including A. actinomycetemcomitans attach to the initial colonizers.
- Adhesion to epithelial cells: Most of the A. actinomycetemcomitans strains bind strongly to the epithelial cells 102. Binding is rapid, which reaches saturation within 1 hour 103. Cell surface entities that mediate adhesion are fimbriae, extracellular amorphous material and extracellular vesicles 102.
- Binding to extracellular matrix proteins: In order to initiate disease, A. actinomycetemcomitans must bind to the extracellular matrix. A major component of the extracellular matrix is collagen 104. The major fibers forming the extracellular matrix are collagen Type I, II, III, V, XI. Less abundant, but equally important are noncollagenous glycosylated proteins, fibronectin, and laminin. Outer membrane proteins are important for binding of A. actinomycetemcomitans to collagen and other proteins but binding is highly specific.
Bacteriocins
These are proteins produced by bacteria that are lethal for other strains and species of bacteria. A. actinomycetemcomitans bacteriocins are active against S. sanguis, S. uberis and A. viscosus 105, 106. Bacteriocins increase the permeability of the cell membrane of the target bacteria leading to the leakage of DNA, RNA and macromolecules essential for growth.
Bone resorption factors
A. actinomycetemcomitans stimulate bone resorption in periodontal tissues by various mechanisms;
- Lipopolysaccharides.
- Proteolysis sensitive factor in micro-vesicles.
- Surface-associated material.
The surface-associated material has recently been identified as the molecular chaperone, GroEL. The chaperone appears to act in a direct way with the major bone-resorbing cell population, the osteoclast 107-109.
Collagenase
As collagen is the most abundant component of the extracellular matrix, its destruction leads to the degradation of the extracellular matrix. A. actinomycetemcomitans produces a collagenolytic protein, which can attack collagen. Collagenase is primarily produced by ……Contents available in the book……..Contents available in the book…………..Contents available in the book…………..Contents available in the book……..
Cytotoxins
Fibroblasts are the most important source of collagen in the extracellular matrix. Cytotoxins act on fibroblasts and inhibit their proliferation. The cytotoxin produced by A. actinomycetemcomitans has been identified as a 50 kDa protein that inhibits DNA synthesis of fibroblasts 112.
Extracellular membrane vesicles
Almost all strains of A. actinomycetemcomitans have these vesicles. Growth conditions alter the formation and morphology of vesicles. These vesicles contain leukotoxins, endotoxins, factors involved in bone resorption activity and bacteriocins 105, 106.
Fc binding proteins
These bind to the Fc portion of an antibody and hence, inhibit phagocytosis. Fc portion also activates the complement system, so all these pathways are blocked.
Leukotoxin
The ability of A. actinomycetemcomitans extracts to cause the death of leukocytes was first shown more than 36 years ago 63, 113. Leukotoxin is a member of RTX (repeat in toxin) family of toxins which are responsible for the pore-forming leukotoxic and hemolytic activity 114. The leukotoxin gene (Ltx A) resides on operon consisting of 4 genes C, A, B and D 115. Ltx B and Ltx D are involved in transporting the toxin to the surface of the cell while Ltx C post-transitionally activates the toxin. The expression appears to be regulated by the presence of oxygen and is induced under anaerobic conditions 116. The mechanism of leukotoxicity 117 includes:
- Membranolytic activity, producing pores in the target cell.
- Phospholipids act as the receptor for the toxin whose activity results in a rapid influx of Ca2+ into the cell.
- Necrosis and apoptosis.
The function of systemic antibodies produced against the leukotoxin produced by A. actinomycetemcomitans has been studied recently in a study 118 which was conducted on Swedish population. The results of the study showed that these antibodies might contribute to limit the systemic effects of the infection. The detailed information regarding the Leukotoxin produced by A. actinomycetemcomitans has been given in some recent articles 119, 120.
Lipopolysaccharides (LPS)
These are endotoxins having the potential to modulate the host responses and contribute to tissue destruction. LPS causes tissue destruction by the following mechanisms 121,
- Stimulation of in vitro bone resorption.
- The production of IL-1, TNF and prostaglandin (PGE2) from macrophages.
- Polyclonal activation of B-lymphocytes.
The bone resorptive activities of LPS 122, 123 are the result of stimulation of PGE2 and IL-1 release from osteoblasts and other cells. Along with this A. actinomycetemcomitans is also known to activate the complement cascade by the alternative pathway, which in turn generates prostaglandins and this is the probable mechanism of bone resorption in case of periodontitis 124.
Immunosuppressive factors
A. actinomycetemcomitans produces proteins that inhibit DNA, RNA and protein synthesis by human lymphocytes. It is believed that it affects immunoglobulin production by activating B-cells that downregulate the ability of B- and T-cells to respond to mitogens. In addition, leukotoxin impairs the ability of lymphocytes to respond to mitogens by inhibiting DNA, RNA, protein, IgG and IgM synthesis 101.
Products that inhibit PMN function
A. actinomycetemcomitans produces low molecular weight compounds that inhibit PMN leukocyte chemotaxis. The inhibitory activity is aborted by treatment with proteinase K, which means that the compounds are proteinaceous in nature. Along with this, A. actinomycetemcomitans has been shown to be capable of inhibiting PMNs from producing some potent antibacterial agents that are gained when the PMNs fuse with lysosomes 101.
Penetration of epithelial cells
Research work has shown that A. actinomycetemcomitans can penetrate the gingival epithelial cells 125,126. Entry of the bacteria into the cell permits them to either transit the epithelial cell barrier or persist and grow in a protected cellular environment. Studies done by Meyer et al. (1997) 127,128 suggest that primary receptor for A. actinomycetemcomitans invasion is transferrin receptor. Invasion through integrins is the secondary pathway of entry. A. actinomycetemcomitans invasion of epithelial cells is a highly dynamic and complex process. It involves the attachment of the microorganism to the host cell with the initiation of some form of signaling, binding to a receptor, entry in a vacuole, escape from the vacuole, rapid multiplication, intracellular spread, exit from the cell and cell to cell spread 128.
Know More…
Clonal types in the pathogenesis of periodontal diseases:
The first step in the pathogenesis of the periodontal disease is the adherence of bacteria to periodontal tissue. Multiple clonal types may exist for a particular bacterial species, all of which may not be similarly virulent. Amano and co-workers (2000) 132 have reported that both disease-associated and non-disease associated genotypes exist in P. gingivalis, suggesting that there is a significant predominance of P. gingivalis with type II fimA in periodontitis patients. Highly leukotoxic clones of A. actinomycetemcomitans have been identified 133. Other studies have demonstrated the production of leukotoxin from A. actinomycetemcomitans is regulated in part by environmental factors, such as atmosphere, nutrition supply, and pH 134-136. The optimal pH for the growth of A. actinomycetemcomitans is reported to be in the range of 7.0-8.0 135.
Cytolethal distending toxin (CDT)
It has been shown that A. actinomycetemcomitans also produces a 60-kDa protein, which downregulates both T- and B-cell responsiveness through the activation of a sub-population of B-lymphocytes 129. The cytolethal distending toxin is encoded by a locus of three genes, Cdt A,B,C 130, 131. It impairs the lymphocyte function by arresting its cell cycle. The active subunit, CdtB, exhibits DNase I activity. While the role of CdtA and CdtC is less clear, both proteins possess putative mucin-like carbohydrate-binding domains that predict interaction with the host cell surface.
Conclusion
A. actinomycetemcomitans is a very important periodontal pathogen. It has been demonstrated in various studies that it plays a very important role in the etiopathogenesis of periodontitis. Most importantly, this bacteria has been associated with Grade C periodontitis. Hence, it is essential that we must have an in-depth knowledge regarding this bacterium. Many aspects regarding A. actinomycetemcomitans are still under investigation and its role in the formation of dysbiotic biofilm still needs a lot of research.
References
References are available in the hard copy of the website.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

