Introduction to bleeding disorders
There are various causes of excessive bleeding including coagulation factor deficiencies, fibrinolytic defects, vascular disorders and platelet disorders. Patients with congenital bleeding disorders have an increased risk of significant bleeding from invasive dental and oral surgery procedures. A thorough knowledge of the blood clotting mechanism is essential for the understanding and management of patients with bleeding disorders. The blood clotting mechanism involves various factors which participate in the formation of blood clot. Coagulation is a property of plasma alone, not of blood cells. Normal clotting time with lee’s white method is 6-17 min. Morawitz in 1904 described the basic mechanism of blood clotting. More than 50 substances that affect blood coagulation have been found in the blood. Most important of these clotting factors are,
I. Fibrinogen
II. Prothrombin.
III. Tissue thromboplastin.
IV. Calcium.
V. Proaccelerin or labile factor.
VI. There is no factor VI.
VII. Serum prothrombin conversion accelerator or stable factor.
VIII. Anti-haemophilic factor (AHF).
IX. Plasma thromboplastin component (Christmas factor).
X. Stuart factor.
XI. Plasma thromboplastin antecedent.
XII. Hageman or surface factor.
XIII. Fibrin stabilizing factor.
In an area where there is restricted blood flow or an abnormal vessel wall without injury, the initiation of clot formation is by the intrinsic pathway. The initiation of fibrin clot formation in response to injury to the tissue is carried out by extrinsic pathway. Both of these pathways converge in a final common pathway involving the conversion of prothrombin to thrombin and thrombin-catalyzed cleavage of fibrinogen to fibrin.
The congenital bleeding disorders include hemophilia A (classic hemophilia) and B (Christmas disease), von Willebrand disease (vWD) and Factor XI deficiency. Both hemophilia A and B are inherited as an X-linked recessive conditions and share identical clinical manifestations. Out of hemophilia A and B, hemophilia A is more common, accounting for approximately 85% of all the cases of hemophilia (incidence, 1:5,000 live male births), and is characterized by the deficiency of factor VIII. Hemophilia B is characterized by the deficiency of factor IX (incidence, 1:30,000 live male births) 87.
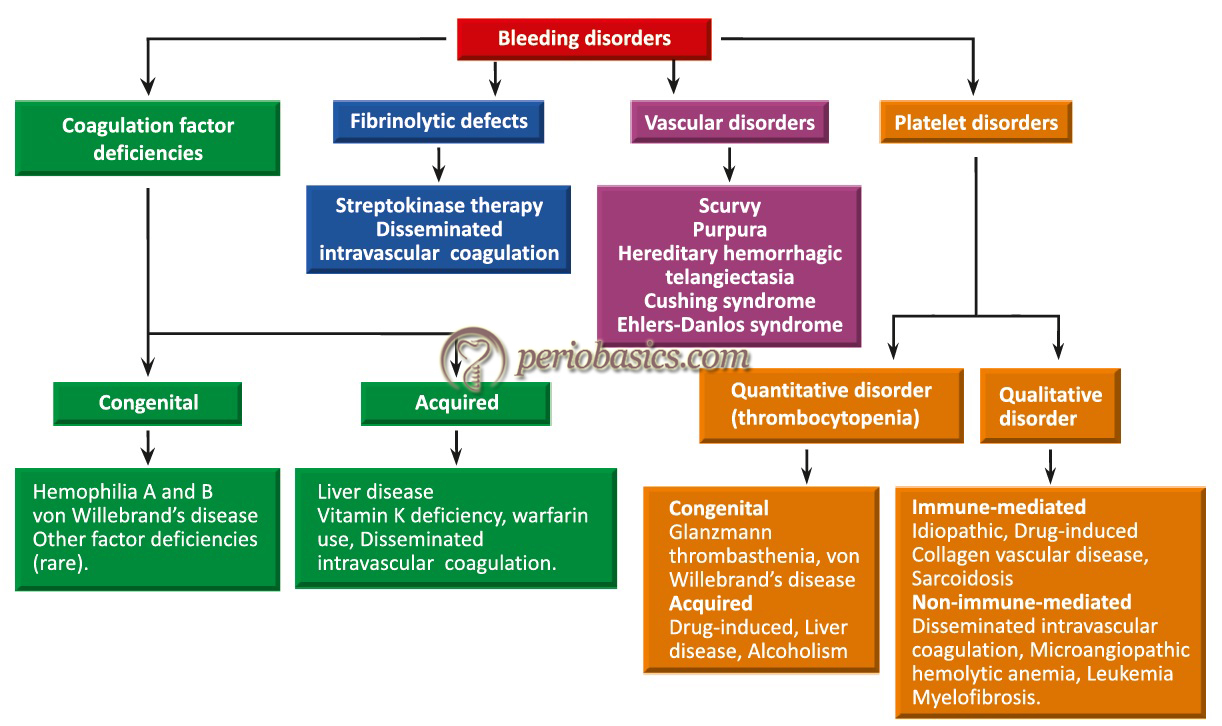
The commonest of the congenital bleeding disorder is vWD and is characterized by a deficient or abnormal plasma protein known as von Willebrand factor (vWF). It is an autosomal dominant condition and affects both males and females. Inherited vWD is subdivided into 3 categories: Types 1 and 3 represent a partial and complete deficiency of vWF, respectively; and Type 2 variants represent qualitative abnormalities of vWF (Table 32.14). The most common type of von Willebrand disease (vWD) is Type 1 vWD, which counts for 80% of all vWD patients 88, 89. The management of vWD patients depends on the type of disease. Most of the patients with Type 1 and some Type 2A and 2M patients respond well to desmopressin acetate, a synthetic analog of vasopressin (explained later). Patients with Type 2B and Type 3 vWD, require vWF replacement. The currently available replacement is vWF containing Factor VIII concentrates derived from pooled human plasma which is pasteurized to inactivate viruses 90-92.
| Classification of von Willebrand disease. | |
|---|---|
| Type 1 | Partial deficiency (80% of those with vWD) |
| Type 2 | Qualitative defects |
| A | Decreased function; absent medium and high-molecular-weight multimers |
| B | Increased affinity for platelet GPIb; decreased high-molecular-weight multimers |
| M | Decreased function; variable multimer pattern |
| N | Decreased affinity for Factor VIII |
| Type 3 | Severe deficiency; autosomal recessive |
Other congenital factor deficiencies include fibrinogen, Prothrombin, Factor V, Factor VII, Factor X, Factor XI and Factor XIII deficiencies. These are relatively rare factor deficiencies and are treated with fresh frozen plasma or specific factor transfusion.
Specific blood tests to confirm bleeding disorders
Following investigation are done in bleeding disorders:
Bleeding time: Time taken for a standardized skin puncture to stop bleeding is calculated. Depending on the method used, it varies from 2-9 minutes. The abnormality is found when there is a defect in platelet number or function.
Platelet count: It is obtained from anticoagulated blood using an electronic particle counter. The normal range is 150 to 450 × 103/mm3. Decreased platelet count is associated with bleeding disorders.
Prothrombin time: It measures the adequacy of the extrinsic and common coagulation pathway. In this test, the tissue factor is replaced by exogenously added source of tissue thromboplastin eg. brain extract. Calcium ions are provided from outside. Increased PT is present in the deficiency of factor V, VII, X, prothrombin, and fibrinogen. It is calculated in seconds.

Partial thromboplastin time: It checks the integrity of intrinsic and common clotting pathway. It is calculated in seconds. Intrinsic pathway requires a contact phase that is provided by the surface of the blood vessels on which surface endothelium is altered. In vitro, it is provided by Kaolin. Cephalin is a substitute for platelet phospho-lipids. The plasma clotting time is checked. Increased PTT indicates a deficiency of factor I, II, V, VIII, IX, XI, XII. Any of these factors may be deficient when PTT is increased.
Activated partial thromboplastin time (APTT): In APTT, an activator is added that speeds up the clotting time and results in a narrower reference range. The APTT is considered a more sensitive version of the PTT.
| Laboratory tests for bleeding disorders. | ||
|---|---|---|
| Test | Normal values | |
| Bleeding time (ivy) | BT | 3-7 minutes |
| Clotting time | CT | 12-15 minutes. |
| Prothrombin time | PT | 11 - 13.5 Sec. |
| Activated Partial Thromboplastin Time | APTT | 30 - 40 Sec. |
| Platelet count | 250,000 ± 150,000/mm3 | |
Periodontal treatment of patients with bleeding disorders
The bleeding disorders are clinically confirmed by the case history of the patient. Usually, most of the patients are under medical supervision for their condition. Most of the procedures in non-surgical periodontal therapy do not require augmentation of coagulation factor levels. Procedures that require increment in the clotting factor levels (depending on the type of hemophilia and vWD), there may be four therapeutic management options,
1. Coagulation factor replacement therapy.
2. Release of endogenous factor stores using desmopressin (DDAVP).
3. Improving clot stability by antifibrinolytic drugs, for example, tranexamic acid.
4. Local hemostatic measures.
| Classification of hemophilia. | ||
|---|---|---|
| Severity | Serum levels of factor | Bleeding severity |
| Mild | 0.05-0.35 IU/mL or 5% to 35% | No spontaneous bleeding; delayed onset bleeding after trauma or surgery or dental extractions |
| Moderate | 0.01-0.05 IU/mL or 1% to 5% | Bleeding into joints or muscles with minor trauma; excessive bleeding with surgery |
| Severe | < 0.01 IU/mL or < 1% | Spontaneous joint, muscle and internal bleeding; excessive bleeding with trauma or surgery |
Coagulation factor replacement therapy
The patient may have mild, moderate or severe hemophilia. In mild hemophilia, there is no need for clotting factor replacement. Agents such as desmopressin may be given intravenously, prior to the surgery, to increase the level of factor VIII and vWB factor in the blood. In the case of moderate and severe hemophilia A and B, coagulation factor replacement therapy is the main form of therapy. The concentrate of the deficient factor is administered by intravenous infusion. The dental treatment should be performed within 30 minutes to one hour after infusion of clotting factors because the level of clotting factor in the blood declines with time. The factor administration may be prophylactic (to prevent bleeding) or may be ………. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Release of endogenous factor stores using desmopressin (DDAVP)
Desmopressin (1‐desamino‐8‐d‐arginine vasopressin) is an analog of vasopressin that exerts a substantial hemostatic effect by inducing the release of vWF from its storage sites in endothelial cells. Vasopressin is an anti-diuretic hormone which functions through two receptors, termed V1 and V2, activating different intracellular second messengers. The agonist activity at V2 receptors leads to the rise in intracellular concentrations of cyclic adenosine monophosphate, which in turn induces exocytosis of vWF from its storage sites (i.e., Weibel-Palade bodies of endothelial cells) into the circulation. It also increases the plasma concentration of factor VIII. Desmopressin being an analog of vasopressin, activates only V2 receptors and thus causes the release of vWF into circulation. It shortens the prolonged APTT and the bleeding time. DDAVP (0.3 μg/kg in 50 ml of normal saline) can be administered intravenously (4 μg/ml concentration) one hour pre-procedure as a slow intravenous infusion over 20-30 minutes. It can also be administrated through subcutaneous or intranasal routes.
Side effects are primarily the result of vasodilation and include flushing, tachycardia, hypotension, and headache. Other side effects include hyponatremia (water intoxication in severe cases) resulting from desmopressin’s antidiuretic effect and thrombosis in patients with pre-existing risk factors.
Antifibrinolytic agents
These agents promote blood coagulation by their anti-fibrinolytic activity. Epsilon-aminocaproic acid (EACA), and the more potent tranexamic acid (TA) are used as anti-fibrinolytic agents. These are analogs of the amino acid lysine. They exert their antifibrinolytic effect by interfering with the binding of plasminogen to fibrin, which is necessary for the activation of plasminogen to plasmin. The oral dose of TA is 15-25 mg/kg, which approximates to 1 g for the majority of adults, every 6-8 hours. Its oral administration should be started two hours pre-operatively and should be continued for up to 7-10 days post-procedure.
TA is freely soluble in water, so 10 ml of a 5% solution TA solution can be used as mouthwash just before the oral surgical procedure and should be continued ………. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………
Local hemostatic measures
In patient with bleeding disorder, the local hemostatic measures can be used to stop bleeding. Placement of sutures and pressure pack is very helpful in stopping the bleeding. Various local hemostatic agents available include oxidized cellulose, Surgicel®, resorbable gelatin sponge, Gelfoam®, cyanoacrylate tissue adhesives and surgical splints. It must be remembered that minimum trauma to tissue during the surgical procedure is the key to quick control of bleeding.

Platelet Disorders
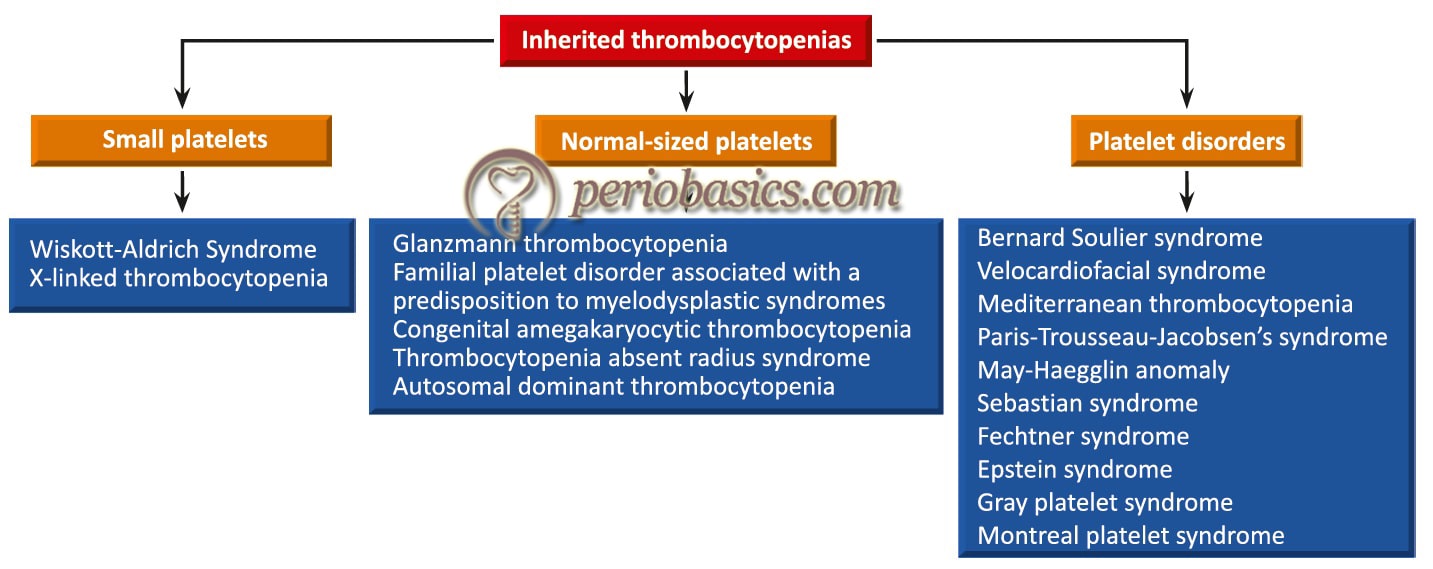
Platelets are one of the most important components of clotting mechanism. The normal platelet count is 250,000 ± 150,000/mm3. If the platelet count is less than 100,000 cells/mm3, the condition is known as thrombocytopenia. When the platelet count is below 50,000 cells/mm3 there is excessive bleeding during the surgery and post-operatively. Many different types of platelet disorders are there which may cause defective coagulation. They are broadly classified into various categories on the basis of defects of platelet number (thrombocytopenia) or their function. Thrombocytopenias may be broadly classified as inherited or acquired, containing different types of thrombocytopenias under these categories.
Idiopathic/immune thrombocytopenic purpura (ITP)
ITP is also known as immune thrombocytopenic purpura or primary immune thrombocytopenic purpura. It is a hemorrhagic disorder characterized by abnormally increased destruction of circulating platelets. In this condition, platelets are opsonized by autoreactive antibodies and prematurely destroyed by the reticuloendothelial system. ITP occurs in two distinct clinical forms: an acute self-limiting form seen almost exclusively in children and chronic form mostly observed in adults. The oral findings in this condition are bleeding mucous membranes and petechiae which can be seen in areas most prone to friction.
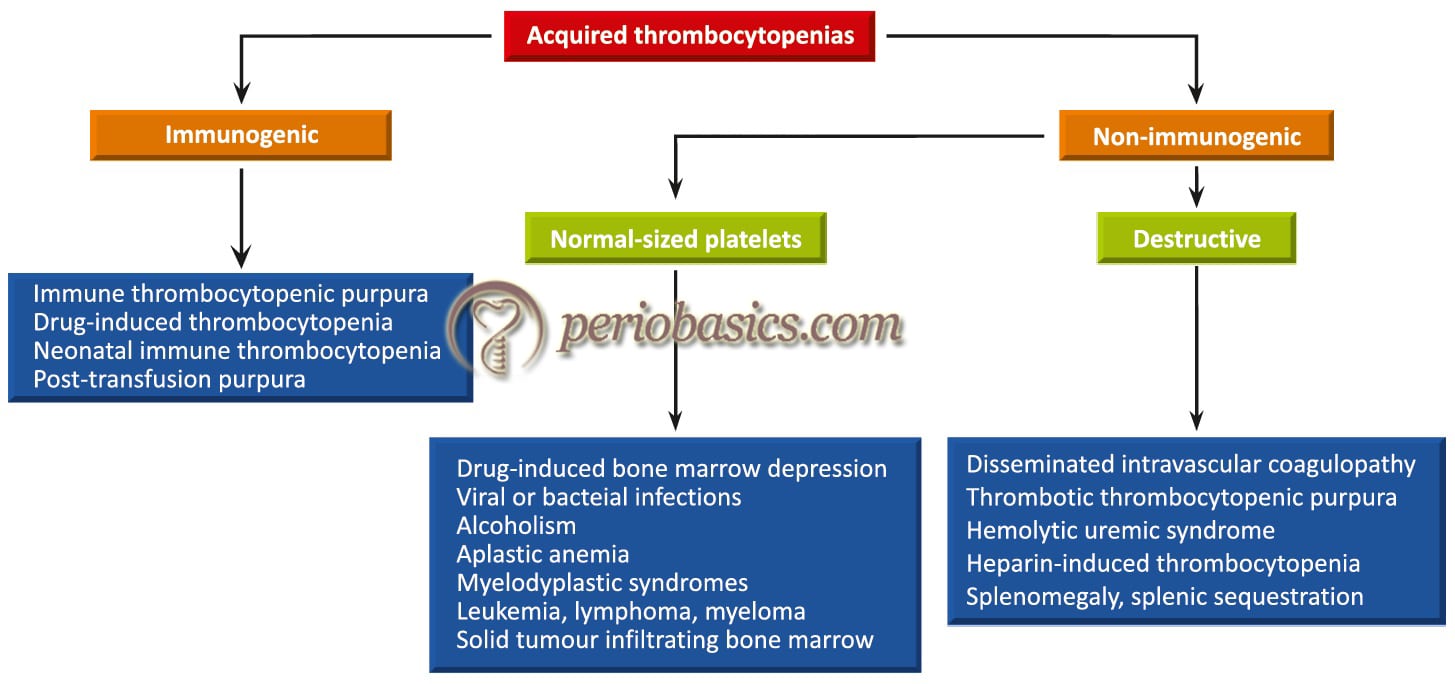
Non-immunogenic thrombocytopenias
In this case, there is no immunogenic destruction of platelets but platelet count is reduced due to either less production or more destruction of the platelet. Platelets may have adhesion (Bernard-Soulier syndrome), aggregation (Glanzmann thrombasthenia) or granule defects (Gray platelet syndrome). Many drugs are also known to have significant adverse effects on platelet number and function. Estimates on the incidence of drug-induced thrombocytopenia range 5-40% in patients receiving heparin, to < 1% with other causative agents 93. Other drugs most frequently associated with the development of thrombocytopenia are quinidine, gold and trimethoprim-sulphamethoxazole 94. Nonsteroidal anti-inflammatory drugs (NSAIDs) attenuate the platelet activity.
The most common of these is aspirin, which acetylates COX, thereby blocking thromboxane A2 (TxA2) release from activated platelets, thus attenuating platelet activity. Periodontal treatment of patients with platelet disorders: A detailed history of the patient should be recorded with a description of the type of platelet disorder and the medical treatment provided to the patient for the disorder. If the platelet count is > 75,000/mm3, there is no need of additional support, but the operator should be prepared to treat prolonged bleeding by using sutures, hemostatic agents, pressure packs, gelatin foams, and so forth. If the platelet count is 40,000 to 75,000/mm3, platelet transfusions may be considered pre- and 24 hours postoperatively.
Localized procedures to manage prolonged bleeding may include sutures, hemostatic agents, pressure packs, and/or gelatin foams. If the platelet count is ………. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………
Blood dyscrasias
The hematological malignancies can be grouped into three categories: leukemia, lymphoma and plasma cell tumors. These malignancies may alter bleeding and clotting time, wound healing and may increase susceptibility to infection. Many patients suffering from hematological disorders seek periodontal treatment. A periodontist should be aware of these disorders as well as the protocol followed during the treatment.
Leukemia
Leukemia is a hematological disorder which is caused by proliferating white blood cell-forming tissues, resulting in a marked increase in circulating immature or abnormal white blood cells. It results from the proliferation of a clone of abnormal hematopoietic cells with impaired differentiation, regulation, and apoptosis (programmed cell death). The multiplication of leukemic cell at the expense of normal hematopoietic cell lines causes marrow failure, depressed blood cell count (cytopenia), and death as a result of infection, bleeding, or both 95. Leukemia is classified based on clinical behavior (acute or chronic) and the primary hematopoietic cell line affected (myeloid or lymphoid). The four principal diagnostic categories are the following 96, 97:
1. Acute myelogenous leukemia (AML),
2. Acute lymphocytic leukemia (ALL),
3. Chronic myelogenous leukemia (CML) and
4. Chronic lymphocytic leukemia (CLL).
Oral findings
Patients with leukemia show gingival bleeding, petechiae and ecchymosis due to thrombocytopenia. The severity of these symptoms depends on the platelet count. Less is the platelet count, more severe are these findings. Another important finding is gingival enlargement. It is more common in acute than chronic leukemia. Gingival enlargement is secondary to infiltration of the gingival tissue with leukemic cells. It is characterized by the progressive enlargement of interdental papillae as well as marginal and attached gingiva. Gingiva appears swollen, devoid of stippling and pale red to deep purple in color. Gingival infiltration by leukemic cells also predisposes the patient with leukemia to bleeding 98. Generally, gingival ………. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Periodontal treatment of patients with leukemia
The consultation with the physician treating the malignancy of the patient is a must prior to starting the periodontal treatment. Once the condition of the patient is understood, treatment is planned for the patient. Dental treatment of a leukemic patient should be done according to the following criteria 98,
- The physician or the oncologist treating the patient should be consulted prior to the initiation of dental treatment.
- A detailed case history, thorough periodontal and dental examination and radiographic examination are essential before starting the treatment.
- Dental treatment should be performed before starting the chemo/radiotherapy.
- Patients in long-term remission can undergo dental treatment, while patients with advanced or relapsed disease with reserved prognosis should receive palliative or urgent treatment only.
The main problems in the dental treatment of patients with hematologic malignancies of white cells are:
1. Tendency to bleed.
2. Increased risk of infection,
- Odontogenic.
- Opportunistic.
3. Anemia.
4. Secondary adrenal insufficiency due to corticosteroids.
Tendency to bleed
As already stated, intraoral bleeding is commonly found in these patients clinically manifested as petechiae and ecchymosis, and occasionally hematoma formation. It is caused due to thrombocytopenia due to suppressed platelet formation. In procedures where bleeding is not anticipated, treatment can generally be provided in even severely thrombocytopenic patients without the need for transfusions. If the surgical procedure has to be performed protocol followed during the treatment of thrombocytopenia (explained previously) is followed.
Increased risk of infection
The risk of infection in leukemic patients is increased due to compromised lymphocyte function or low neutrophil count. The patient may present with an oral infection, which may be of odontogenic origin or opportunistic.
Odontogenic infections
Because of compromised immune system, the patient is more prone to have pulpal or periodontal infections. It has been shown that oral prophylaxis, oral hygiene instructions and elimination of oral sources of infection before the initiation of cancer treatment, can significantly reduce the risk of infectious complications 102, 103. The periodontal treatment should be given after evaluation of the platelet (for bleeding tendency) and absolute ………. Contents available in the book……… Contents available in the book……… Contents available in the book……… Contents available in the book………
Opportunistic infections
These patients are also at a higher risk of developing opportunistic infections like viral and fungal infections. The viral infection may be primary or due to reactivation of the latent virus. Herpes simplex virus (HSV) is the most common viral infection in these patients and typically presents as single or multiple painful ulcerative lesions that may involve any oral mucosal surface. Antiviral therapy is given to treat these infections.
The fungal infection by Candida spp. is common during intensive chemotherapy and in advanced disease. Candidiasis can present as pseudomembranous (most common), erythematous, hyperplastic, or angular cheilitis. The patient is given a topical application of nystatin for 7 to 14 days. In severe cases, 100 to 200 mg/day of fluconazole or itraconazole is given systemically for 7 to 14 days.
Anemia
The patient may have anemia due to decreased Hb. Patients with severe anemia often complain of easy fatigue and inability to tolerate time-consuming dental treatment. Patient’s oncologist should be consulted regarding this problem and only after appropriate treatment, long dental procedures should be planned.
Secondary adrenal insufficiency due to corticosteroids
Patients with leukemia are usually on corticosteroid therapy, so there are chances of development of secondary adrenal insufficiency. The dental treatment of these patients should be done only after thorough evaluation of the patient and discussion with the patient’s physician. If during the treatment, the patient shows signs and symptoms of adrenal insufficiency, the treatment protocol is the same as discussed for adrenal crisis in the previous section.
Conclusion
Many patient suffering from various hematological conditions seek dental treatment. As many periodontal treatments involve bleeding, it is essential for periodontists to understand various hematological disorders and the periodontal management of patients with these disorders. In certain situations, it is recommended to consult the physician before rendering the periodontal treatment.
References
References are available in the hard-copy of the website.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

