Introduction to osteoimmunology
Periodontal arena harbors a very complex biofilm which consists of numerous bacterial species 1, 2. As this biofilm matures, there is an increased accumulation of facultative anaerobic, Gram-negative microorganisms 3, which result in early vascular changes in the periodontium, with exudation and migration of phagocytic cells, including neutrophils and monocytes/ macrophages, into the junctional epithelium and gingival sulcus, causing initial gingival inflammation. The most important and most prevalent anaerobic Gram-negative bacteria in the subgingival arena are Aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis, Prevotella intermedia, and Tannerella forsythensis. These bacteria play a crucial role in periodontal connective tissue destruction, and alveolar bone resorption by means of an immunopathogenic mechanism, participating in the periodontal pocket formation and subsequently, development of periodontitis. Once periodontitis has been established, an inflammatory infiltrate is formed consisting of different kinds of cells, such as macrophages and lymphocytes that will produce different cytokine subtypes, which are responsible for the disease progression.
Gingival sulcus is the site for initiation of host-microbial interactions. The inflammatory cells, including neutrophils, macrophages, monocytes, etc., produce many chemical mediators that lead to various immunopathological processes. Cytokines induced by the host response play a critical role in periodontal tissue breakdown 4-7. The periodontal bone loss is the result of the extension of inflammation in the periodontal tissues.
It must be noted here that ……… Contents available in the book……….. Contents available in the book……….. Contents available in the book……….. Contents available in the book……….. Contents available in the book…..
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Mechanism of physiological bone resorption
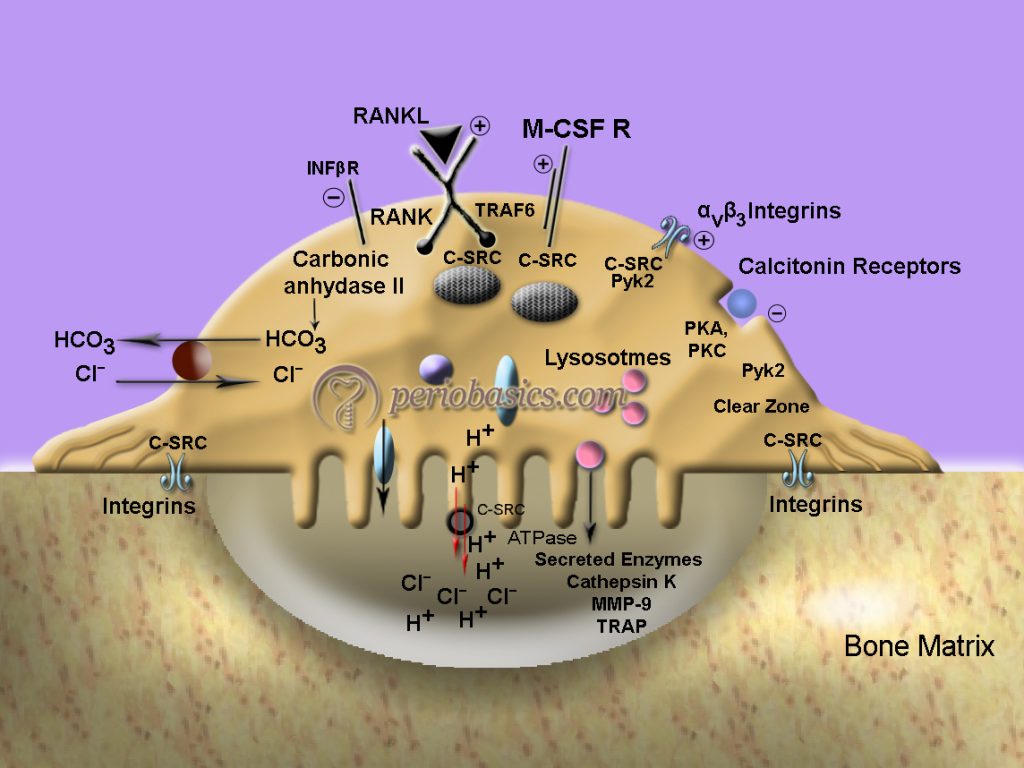
To understand the bone resorption in periodontal diseases, first, let us understand physiological bone resorption. The osteoclasts typically contain multiple copies of the Golgi complex, organized as rings surrounding each nucleus, and many readily identifiable transport vesicles moving toward the ruffled border membrane. The enzymes are secreted via the ruffled border, into the extracellular bone-resorbing compartment, where they reach a sufficiently high extracellular concentration that allows extracellular de-gradation of the bone matrix, within the sealed compartment. The transport and targeting of these secreted enzymes at the apical pole of the osteoclast involves mannose-6-phosphate receptors. Furthermore, the cell secretes several metalloproteinases, including collagenase and gelatinase which are responsible for the breakdown of the matrix. It occurs in the following steps:
- After getting a signal, the osteoclast precursor becomes activated and becomes a multi-nucleated cell, osteoclast.
- Osteoclast now attaches to the bone matrix. The attachment of the cell to the matrix is performed via integrin receptors (mostly αvβ3, αvβ5, and α2β1), which bind to specific sequences in matrix proteins, and activate a specific signaling cascade requiring several signaling molecules to ensure adhesion and cell motility (c-Src, Pyk2, Cbl, Gelsolin).
- There are deep foldings of the plasma membrane in the area facing the bone matrix, called as the ruffled border, which is formed as a result of the directional insertion of the vesicles required for active secretion of protons and lysosomal enzymes toward the bone surface, as well as directional endocytic activity from the bone-resorbing compartment.
- The area of the interface between the cell and the bone surface is surrounded by a ring of contractile proteins (sealing zone) that serves to attach the cell to the bone surface while ensuring its continued ability to migrate.
- The basolateral plasma membrane of the osteoclast is highly and specifically enriched in Na+/K+ ATPase (sodium-potassium pump), HCO3/Cl- exchangers, and Na+/H+ exchangers, as well as several ion channels.
- This membrane also expresses RANK, the receptor for RANK ligand (discussed later) and the M-CSF receptor, all of which are responsible for osteoclast differentiation, as well as the calcitonin receptor, which is capable of rapidly inactivating the osteoclast.
- The osteoclast acidifies the extracellular compartment 12 by secreting protons across the ruffled border membrane, a process that involves the transporting activity of vacuolar proton pumps 13.
- The protons are provided to the pumps by carbonic anhydrase, an enzyme that is highly concentrated in the cytosol of the osteoclast.
- ATP and CO2 are provided by the mitochondria, which are characteristically found in very high numbers in the osteoclast.
- Apical chloride channels (ClC-7) are also found in the ruffled border membrane 14, where they serve to prevent the hyperpolarization created by the massive extrusion of positively charged protons by the V-ATPase.
- First, the hydroxyapatite crystals are mobilized by digestion of their link to collagen (the noncollagenous proteins) and dissolved by the acidic environment. Then, the residual collagen fibers are digested by the specific action of cathepsin K at low pH, and possibly by the activation of latent collagenase.
- During active resorption, the degraded bone matrix is processed extracellularly, after which some products are endocytosed into the osteoclast and degraded within secondary lysosomes, and others are transported through the cell by transcytosis and secreted through the basal membrane.
- The degraded products from the bone matrix may also be released from the bone matrix into the local micro-environment during the periods of relapse of the sealing zone.
Mechanism of bone destruction in periodontal diseases
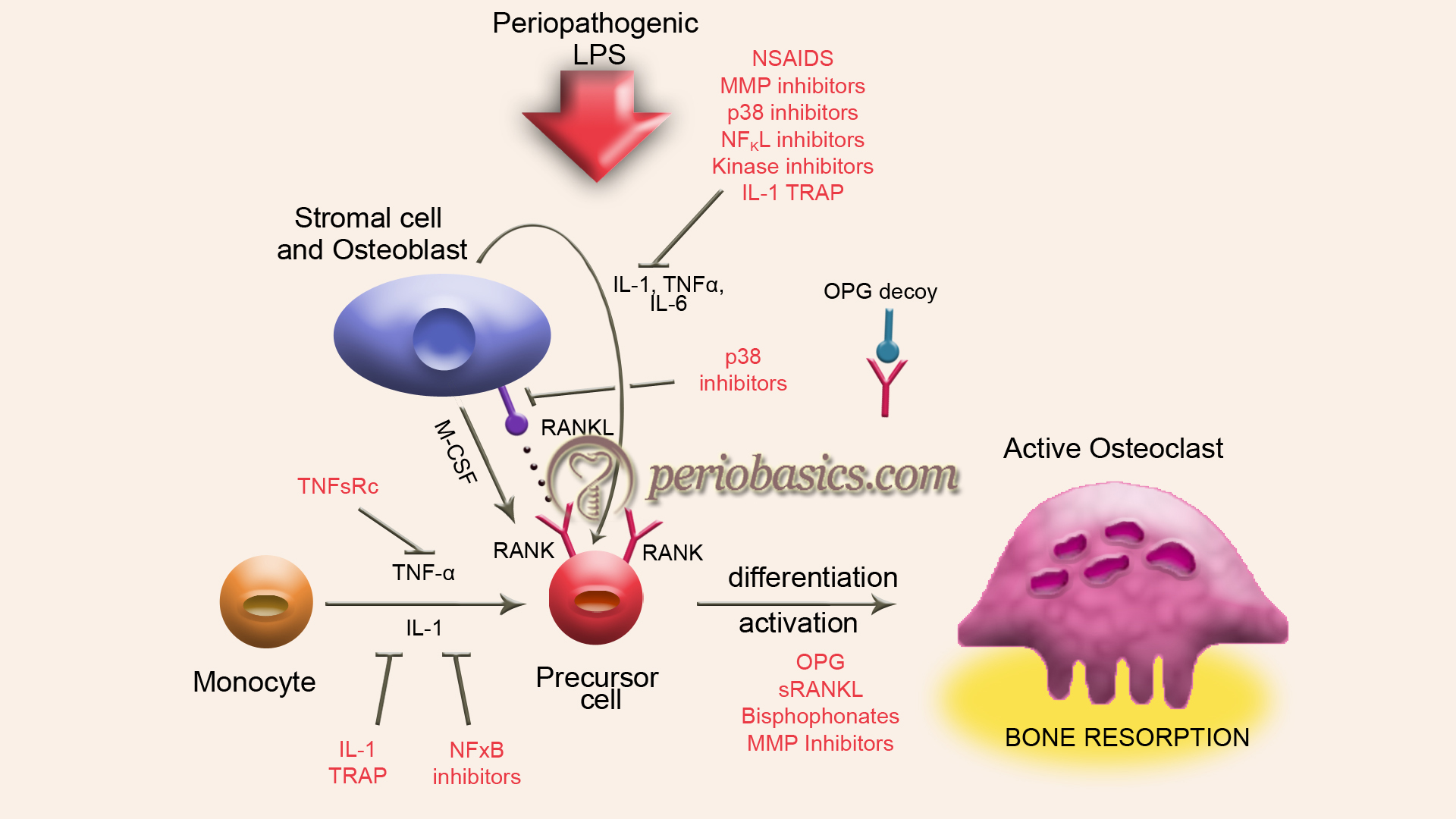
The microorganisms that are present in a biofilm have been shown to be the primary etiological factors for periodontal bone destruction 15. But, now it is clear that the host immune responses to these microorganisms are responsible for the destruction of periodontal bone tissues by the production of RANKL from activated lymphocytes that react to such periodontal pathogens 16, 17.
The identification and characterization of RANKL, its receptor RANK, and soluble decoy receptor osteoprotegerin (OPG) have significantly contributed to the understanding of the skeletal remodeling mechanism involving differentiation of osteoclasts (osteoclastogenesis) and their activation 18-20. In the following paragraphs, we shall try to understand the mechanism of bone resorption in periodontal diseases.
Role of RANKL/ RANK/OPG system and cytokines in bone remodeling
Bone remodeling is a cyclic process of bone matrix synthesis/degradation, rigorously controlled by the RANKL/ RANK/OPG system together with other factors of bone metabolism. A number of hormones and cytokines modulate osteoclastogenesis by enhancing osteoclast differentiation, activation, lifespan, and function. These include parathyroid hormone (PTH), calcitriol, PTH-related protein, prostaglandin E2 and thyroxine 21, 22.
Several pro-inflammatory cytokines have been identified as key molecules contributing to the destruction of periodontal tissue, including interleukin-1 (IL-1), tumor necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ), interleukin-6 (IL-6) 23. Pro-inflammatory cytokines IL-1 and TNF-α have been shown to be very important factors in periodontal bone loss 24-26, and there is evidence that osteoclasts can be activated in an RANKL independent pathway via these pro-inflammatory cytokines 27-29. TNF-α and IL-1 engage initially distinct signaling pathways that converge with activation of the transcription factor, nuclear factor kappa B (NfκB), and stimulation of the mitogen-activated protein kinase (MAPK) system. The combined effect of ……… Contents available in the book……….. Contents available in the book……….. Contents available in the book……….. Contents available in the book……….. Contents available in the book…..
RANKL
RANKL is an important factor associated with bone metabolism. It is also known as receptor activator of NFκB ligand or OPGL (osteoprotegerin ligand) or ODF (osteoclast differentiation factor) or TRANCE (TNF-related activation-induced cytokine). It was cloned and identified through different strategies by four independent groups of researchers: Yasuda 31, Lacey 32, Anderson 33 and Wong 34. RANKL belongs to the TNF family and is the only cytokine which has a role in the development and activation of osteoclasts 35-37. RANKL is a trimeric molecule that can be membrane-associated or soluble molecule released through enzymatic cleavage by membrane metalloproteinases. The proteolytic cleavage of RANKL is carried out by matrix metalloproteinases (MMP3 or MMP7) 38 or ADAM (a disintegrin and metalloprotease domain) 39.
Sources:
Osteoblasts,
Bone marrow stromal cells,
Chondrocytes,
Activated T-lymphocytes.
Biological effects of RANKL
RANKL mediates osteoclastogenesis i.e. it is involved in bone resorption. Activation of mature osteoclasts exclusively depends on RANKL, which in turn are the main effector cells of bone resorption. Gene knockout mice deficient in RANKL or RANK, respectively, not only present osteopetrotic phenotypes but also lack peripheral lymph nodes 40, 41. RANKL also interacts with osteoprotegrin which is an antagonist to osteoclast action. Thus, RANKL has a dual antagonistic type action on osteoclastogenesis, depending on the type of receptor it interacts with i.e. RANK or OPG, although both receptors belong to the same TNF receptor family.
RANK
RANK (receptor activator of NFκB) is also known as TRANCE-R. It is a heterotrimer, expressed in a transmembrane way on the surface of osteoclast progenitor cells, mature osteoclasts, chondrocytes, dendritic cells, trophoblasts and epithelial cells of mammary glands. When hematopoietic stem cells differentiate from the colony-forming unit for granulocytes and macrophages (CFU-GM) to the colony-forming unit for macrophages (CFU-M), the macrophage colony-stimulating factor (M-CSF) induces the expression of RANK on CFU-M. Subsequently, RANK remains expressed on osteoclast lineage cells throughout their lifespan until terminally differentiated into multinucleated cells 42. The interaction of the RANK receptor with its ligand RANKL represents the ……… Contents available in the book……….. Contents available in the book……….. Contents available in the book……….. Contents available in the book……….. Contents available in the book…..
Osteoprotegerin(OPG)
Its bone protective action justifies the name of osteoprotegerin. OPG is secreted by many cell types in addition to osteoblasts, including those in the heart, kidney, liver, and spleen. It is also called as OCIF (osteoclasts inhibitor factor). Osteoprotegerin functions as a soluble decoy receptor, which binds to RANKL, through competition with the RANK receptor. Osteoprotegerin has an anti-osteoclastogenic effect through its role as the antagonistic endogenous receptor which, after binding to RANKL, inhibits osteoclast maturation and activation via RANKL and blocks bone resorption 45, 46. OPG expression is regulated in osteoblasts not only by a variety of cytokines but also by some hormones and growth factors 47.
RANKL/OPG ratio in periodontitis
Research has been done to find out and examine the expression pattern of RANKL and OPG in periodontal bone destruction in periodontitis 48-50. The ratio of RANKL/OPG is critically involved in regulating and directing osteoclastogenic and/or osteoblastogenic development 51. Studies have been done on periodontitis patients, where tissue homogenate isolated from diseased periodontal lesion with bone resorption showed significantly higher levels of RANKL protein than gingival tissue taken from healthy subjects, whereas OPG protein detected in diseased tissue was lower than that in healthy gingival tissue 52-53.
In another study, gingival crevicular fluid (GCF) samples from patients with gingivitis demonstrated an elevated RANKL/OPG ratio as compared to healthy subjects 54. Another study showed that ……… Contents available in the book……….. Contents available in the book……….. Contents available in the book……….. Contents available in the book……….. Contents available in the book…..
Role of cytokines in osteoimmunology
As already stated, cytokines play an important role in the initiation of bone loss in periodontitis. IL-1β and TNF-α are the key players in this process but along with these, there are many other cytokines which are directly or indirectly involved in causing alveolar bone loss in periodontitis. Let us try to understand the mechanism of action of these cytokines,
IL-1
As already discussed, IL-1 is a proinflammatory cytokine. It exists in two forms which have agonistic activity, IL-1α, and IL-1β. For control purposes, there is a third ligand called IL-1 receptor antagonist (IL-1RA) that functions as a competitive inhibitor. IL-1α and IL-1β exert their effect by attaching to cell surface receptors, designated IL-1 receptor-1 (IL-1R-1) and IL-1 receptor-2 (IL-1R-2). IL-1R-1 is thought to mediate most of the functions of IL-1 and IL-1R-2 functions mostly as a decoy receptor 58. The role of IL-1 in bone resorption stimulated by periodontal pathogens has been comprehensively studied in both human and animal models. It has been demonstrated that gingival crevicular fluid (GCF) levels of IL-1β are elevated at sites of recent bone and attachment loss 59, 60. Garlet et al. (2006) 61 have demonstrated that expression of MMP-1, -2 and -9 and RANKL was correlated with the expression of IL-1 β in a time period characterized by the intense increase of the inflammatory reaction and alveolar bone loss. In another study, Mizutani et al. (2013) 62 showed that in IL-1R antagonist (IL-1Ra) knockout mice stimulated with A. actinomycetemcomitans LPS, osteoclast formation, expression of RANK, EP4, and Cox2 mRNAs and production of PGE2 were significantly increased. These studies suggest that IL-1 plays a very important role in the alveolar bone destruction in periodontal disease.
Tumor necrosis factor (TNF):
TNF also exists in two forms TNF-α and TNF-β, exerting its effects by attaching to two structurally similar TNF cell surface receptors, TNF receptor-1 (TNFR-1) and TNF receptor-2 (TNFR-2). Both of these receptors initiate different intracellular signaling cascades and have different cytoplasmic domains. Most of the inflammatory effect is mediated through TNFR-1 signaling, while TNFR-2 attenuates the inflammatory response induced by TNF 63. TNF-α plays a very important role in alveolar bone loss in periodontitis. It has been shown that levels of TNF-α in GCF are elevated at sites where bone and attachment loss have just occurred 64, 65. In the aforementioned study 62, it was observed that TNFR-1 deficient mice had lower levels of inflammatory cytokines TNF-β, IFN-γ, and RANKL in gingival tissues. Furthermore, the expression of MMP-1, -2, and -9 and ……… Contents available in the book……….. Contents available in the book……….. Contents available in the book……….. Contents available in the book……….. Contents available in the book…..
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Interferon-γ (IFN-γ):
As discussed in “Cytokines and their role in pathogenesis of periodontal diseases”, IFN-γ is produced by activated T-cells and NK-cells that play an important role in host defense mechanisms by exerting pleiotropic activities on a wide range of cell types. It exerts its effects by attaching to its receptor, IFN-γR. It is also an important cytokine involved in alveolar bone resorption in periodontal disease. It has been demonstrated that IFN-γ knockout mice showed a decreased bone loss in response to P. gingivalis infection as compared to controls 7. Another study showed that T-cells extracted from periodontitis patient expressed more IFN-γ compared to T-cell from healthy controls 67. Teng et al. (2005) 68 demonstrated that IFN-γ was associated with enhanced alveolar bone loss mediated by RANKL expressing CD4 Th cell in response to A. actinomycetemcomitans during the progression of periodontal disease.
IL-17:
The Th17 cells express RANKL on their surface; however, they may not activate osteoclasts by a RANKL-RANK interaction. These cells may participate indirectly in alveolar bone resorption by releasing IL-17. However, the research on IL-17 has given us contradictory results. Some clinical research is in favor of IL-17 induced bone loss, whereas some research is in favor of the protective role of IL-17 in bone loss. In a clinical study, IL-17 expression was significantly higher in periodontal lesions, especially in the tissue adjacent to bone destruction, than in control sites 69. On the other hand, a study on IL-17 receptor-deficient mice examined the role of IL-17 in inflammatory bone loss induced by the oral pathogen P. gingivalis. These mice showed enhanced periodontal bone destruction, suggesting a bone-protective role for IL-17 70. Thus, more studies on humans are required to find out the exact role of IL-17 in pathogen-induced alveolar bone loss.
Conclusion
Recent studies have highlighted the role of RANK/RANKL /OPG and pro-inflammatory cytokines in the periodontal bone resorption. Host modulation therapies are also being investigated in this direction to minimize the periodontal connective tissue loss. We need to further investigate and analyze the host-microbial interactions and modulation of host response to minimize the periodontal destruction.
References
References are available in the hard-copy of the website.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

