Introduction to evidence-based practice
Evidence-based practice is defined as the conscientious, explicit, and judicious use of current best evidence in making decisions about the care of individual patients 1. Evidence-based practice involves integrating individual clinical expertise with the best available external clinical evidence from systematic research 2. Specifically speaking about Periodontology, every year, thousands of articles are published in various research journals. This research needs to be systematically involved in routine clinical practice of periodontics. Evidence-based Periodontology is an effective tool to support our decision making in clinical practice. However, we must know how to evaluate the research for its nature, strength, and weakness. In the present chapter, we shall discuss all these aspects of evidence-based Periodontology.

Historical background
Traditionally, the decision-making model in clinical practice was made up of personal clinical experience of the practitioner and the information obtained from other expertise (known to that particular practitioner) in the field and textbooks. However, with the increase in the quantity of research published, it became essential to incorporate that information in the decision-making process. The utilization of medical literature in decision making in medical practice was pioneered at McMaster University, Ontario, Canada, in the 1980s. This new dimension of literature-based problem solving was termed as “evidence-based medicine” (EBM) 3. EBM has been defined as “the integration of the best research evidence with our clinical expertise and our patient’s unique values and circumstances.” 4.
Initially, the database regarding medical research was scattered and the definition of high-quality evidence was also ill-defined. With the introduction of newer research methodologies (such as randomized controlled trials) and statistical tools for synthesizing and analyzing the evidence (systematic reviews and meta-analysis), they became important components of EBM. The revolutionary step in the growth of EBM was the generation of electronic database because of which a large amount of research could be accessed within a short duration of time 5.
Another term to be understood here is evidence-based decision making (EBDM). It refers to searching and interpreting the best scientific evidence and its incorporation in the decision making process in conjunction with clinician’s experience and judgment, the patient’s preferences and other patient-related factors. However, it should be remembered here that the best available evidence is just an adjunct to the clinical expertise of the clinician. Hence, it should never replace the traditional decision-making procedures, but just acts as an adjunct.
Soon after its introduction, the EBM was widely accepted by the medical professionals and similarly by the dental professionals. The American Dental Association (ADA) has defined evidence-based dentistry (EBD) as “an approach to oral health care that requires the judicious integration of systematic assessments of clinically relevant scientific evidence, relating to the patient’s oral and medical condition and history, with the dentist’s clinical expertise and the patient’s treatment needs and preferences.” 6. The next important step in the evolution of evidence-based dentistry was ………..Contents available in the book……….Contents available in the book……….Contents available in the book……….Contents available in the book……….
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Goals of evidence-based dental practice:
The evidence-based dental practice has mainly two goals,
1. Gathering the best evidence/research, and
2. Transferring this knowledge to clinical practice.
Know More…
Differences between evidence-based Periodontology and traditional Periodontology:
1. In evidence-based Periodontology, the best evidence available is used, whereas the basis for evidence in traditional Periodontology is unclear.
2. In evidence-based Periodontology, systematic appraisal of the quality of evidence is done, whereas in the traditional Periodontology appraisal of the quality of the evidence is either unclear or absent.
3. The procedure in evidence-based Periodontology is more objective, more transparent and less biased whereas, it is more subjective, opaque and biased in traditional Periodontology.
4. The evidence-based method is scientifically more sound and widely accepted as compared to the traditional methods.
Application of evidence-based methods
The four fundamental phases of the application of evidence-based methods are,
1. Converting information needed into a clinical question, (Asking)
2. Searching for the best evidence, (Acquiring)
3. Reviewing and critically appraising the evidence, (Appraising), and
4. Applying this information in a way to help the clinical practice. (Applying)
Carr and McGivney (2000) 7 suggested an additional phase, that is,
5. Evaluation and performance of the techniques, procedures or materials. (Assessing)
These phases can be remembered as 5As, that is, Asking, Acquiring, Appraising, Applying and Assessing.
Converting information needed into a clinical question (Asking)
The first step in evidence-based dental practice is starting with a clear and focused question. The information needed should be converted into a clinical question which should be clearly relevant to the problem and should facilitate effective research. The questions are usually related to the diagnosis, pathogenesis, prognosis and treatment planning. For example, we need to find out the evidence regarding the outcome of the non-surgical periodontal therapy in Grade B periodontitis patients. Now, non-surgical ………..Contents available in the book……….Contents available in the book……….Contents available in the book……….Contents available in the book……….
Straus et al. (2011) 4 have described four components of a well-built question describing them as ‘PICO’ process of asking questions. A well-built question should be directly relevant to the question at hand. It should be focused, well-articulated and should facilitate searching of a precise answer. The question should contain all four PICO elements.
It should be noted here that the questions can be of two types: background questions and foreground questions. The background questions generally have two parts. The first part is called as question root (who, what, when, how or why) and the second part refers to any disease, treatment of any other aspect of health care. For example, ‘What is the treatment for Grade C periodontitis?’ In this question, ‘what’ is question root and Grade C periodontitis is the disease. The answer to questions like this is best obtained from textbooks or clinical database. The foreground questions are specific knowledge questions. These are the questions which directly affect clinical decision making. For example, ‘What is the outcome of periodontal regenerative therapy in Grade C periodontitis patients?’ The answers to these questions are best obtained from searching the dental literature and are best suited for ‘PICO’ model. The ‘PICO’ model has two additional considerations: What type of information we want, that is, do we want to know about the diagnosis, etiology/harm, therapy, prognosis or prevention.
| The P.I.C.O. elements for Clinical Questions | ||
|---|---|---|
| P | Patient, Population, or Problem | How would I describe a group of patients similar to mine? |
| I | Intervention, Prognostic Factor, or Exposure | Which main intervention, prognostic factor, or exposure am I considering? |
| C | Comparison or Intervention (if appropriate) | What is the main alternative to compare with the intervention? |
| O | Outcome you would like to measure or achieve | What can I hope to accomplish, measure, improve, or affect? |
| What Type of question are you asking? | Diagnosis, Etiology/Harm, Therapy, Prognosis, Prevention | |
| Type of Study you want to find | What would be the best study design/methodology? |
|
Secondly, to find the best answer to the question in hand what should be the best study design or methodology? The best evidence available presently is obtained from systematic reviews/ meta-analysis/randomized control trials (described later). It depends on the question we are asking, what best evidence we can get. Certain questions can have case-control studies at best while others may have systematic reviews/ meta-analysis/randomized control trials which are the highest level of evidence. Table 94.2 describes the search strategies for different type of search questions.
Let us try to understand the PICO model by ………..Contents available in the book……….Contents available in the book……….Contents available in the book……….Contents available in the book……….
Searching for the best evidence (Acquiring)
While searching for the evidence, we must be able to differentiate between low quality and high-quality research (Levels of evidence). Not all therapeutic recommendations are based on evidence of equal quality. A simple grading system has been suggested to describe the quality of research which is based solely on the design of the clinical study. In this grading scheme, scientific evidence can range from level 1, which is the most scientifically valid, to level 5, which is the weakest form of evidence.
Level 1:
This level consists of clinical practice guidelines, high-quality systematic reviews and meta-analysis. The clinical practice guidelines are systematically developed statements to assist clinicians and patients about appropriate healthcare for specific clinical circumstances 8. These guidelines are based on information obtained from best available scientific evidence such as high-quality systematic reviews and meta-analysis. A panel of experts analyses this evidence and formulates the guidelines in an objective manner. These guidelines may be updated over time as the evidence evolves. An example of updating the clinical practice guidelines over time is the antibiotic prophylactic regimen for the prevention of infective endocarditis proposed by the American Heart Association. The original guidelines were published in 1955 and these were updated many times with the latest recommendation given in the year 2007.
Search strategies for different type of search questions.
| Type of question | Type of study/methodology | MEDLINE filters |
|---|---|---|
| Diagnosis | Controlled trials | Sensitivity and specificity [MH] Diagnosis [SH] |
| Therapy | Double-blind RCTs, meta-analysis and systematic reviews | Randomized controlled trials and meta-analysis [PT] |
| Prognosis | · Cohort studies · Case-control studies · Case series | Experimental cohort studies · Prognosis [MH] · Survival analysis [MH] Morbidity · Mortality [MH] |
| Etiology | · Cohort studies · Case-control studies · Case series | Experimental cohort studies [MH] Risk [TW] |
| Prevention | Randomized controlled trials Cohort studies | Randomized controlled trials [PT], Cohort studies [MH], Prevention and control [SH]. |
| Quality improvement | Randomized controlled trials | Randomized controlled trials [PT], Practice guidelines [PT], Consensus development conference [PT]. |
| (Abbreviations: MH=Medical Subject Heading; PT=Publication Type; SH=Subheading; TW=Text Word) |
||
Let us try to understand now; what are systematic reviews and meta-analysis? A ‘systematic review’ is a form of research that provides a summary of medical reports on a specific clinical question, using explicit methods to search, critically appraise, and synthesize the world literature systematically 9. In simple words, we can say that a systematic review is a multistage process where firstly, we identify all available and reliable publications regarding a specific problem. Then we evaluate the reliability of these publications and compile the available data in a quantitative (meta-analysis) or qualitative manner. A ‘meta-analysis’ refers to ………..Contents available in the book……….Contents available in the book……….Contents available in the book……….Contents available in the book……….
Another component of level 1 evidence is a high quality randomized controlled trial (RCTs) or prospective study. These RCTs are associated with low study errors (low P-value, confidence interval which excludes 1, or a ”negative” result associated with a power of over 80%).
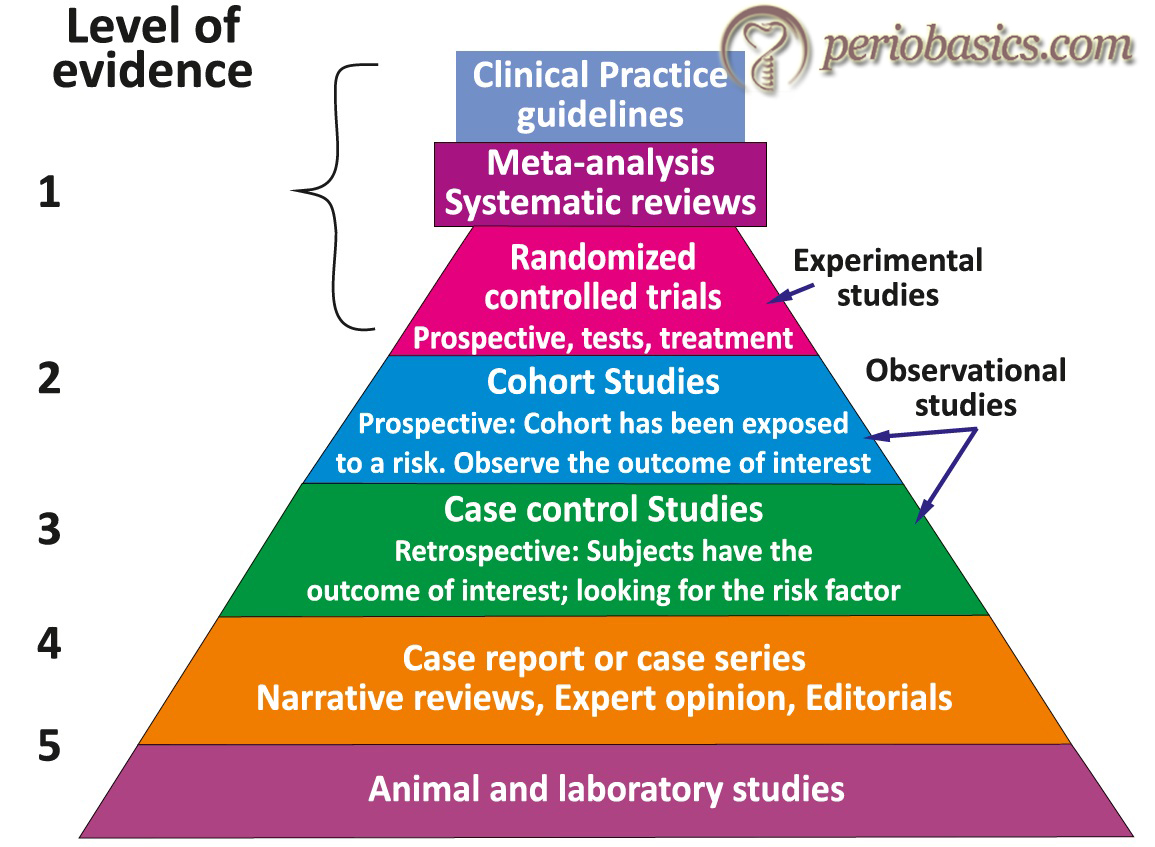
Level 2:
The research work included in level 2 evidence is basically “underpowered” research, that is, not enough patients may have been enrolled in the study to detect a significant difference. Level 2 evidence is also derived from randomized clinical trials, however, these are of lesser quality. These trials compare the outcome of those in the treatment group to those who receive no treatment and the treatment allocation in these trials is random in nature. The RCTs are labeled as level 2 evidence when they are associated with unacceptably high Type I and Type II error. Other research works included in level 2 evidence are prospective comparative study and retrospective study.
Level 3:
Level 3 evidence is derived from nonrandomized, controlled clinical trials. In these studies, the researcher compares the outcome of a particular treatment on a test group of patients and compares that to the control group. However, the reader is unsure of whether the outcome differences are caused by the treatment or some other factor which was not balanced between the two groups. The examples of level 3 research include case-control study (therapeutic and prognostic studies), retrospective comparative study and systematic review of Level 3 studies.
Level 4:
Level 4 evidence consists of case series; case-control study (diagnostic studies); poor reference standard and analysis with no sensitivity analysis. In case-series, the researcher reports on how a series of patients respond to a given intervention. However, the reader ………..Contents available in the book……….Contents available in the book……….Contents available in the book……….Contents available in the book……….
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
Level 5:
This is the weakest level of evidence that is derived from interventional case reports and expert opinion. In interventional studies, a single patient is subjected to a particular treatment and the results are reported. These studies are usually used to report a novel treatment. The primary limitation of these studies is that the reader is unsure, whether the results of the treatment will be the same or different in other patients subjected to the treatment.

How to search for the evidence?
The sources of the evidence may be primary or secondary. The primary sources of evidence include original research studies and publications that have not been filtered or synthesized. For example, a randomized controlled trial or a cohort study. On the other hand, the secondary sources are synthesized studies and publications of the already conducted primary research. These include systematic reviews, meta-analysis and evidence-based article reviews. It should be remembered here that the secondary sources of evidence such as systematic reviews and meta-analysis are considered superior to the primary sources such as an individual RCT.
Presently, with the wide availability of the internet, the most common method of searching evidence is the electronic database. There are many excellent and highly specialized databases available today, including MEDLINE, ………..Contents available in the book……….Contents available in the book……….Contents available in the book……….Contents available in the book……….
MEDLINE is most commonly used for the database regarding health-related research. This multipurpose database has been created and maintained by the United States National Library of Medicine (NLM) of the National Institutes of Health. Created in 1966, it is an excellent index to biomedical literature, covering the disciplines of medicine, dentistry, nursing, veterinary medicine, health care services, and the preclinical sciences. At present, this database contains more than 24 million references to journal articles. It contains citations from more than 5,200 worldwide journals in about 40 languages; about 60 languages for older journals. More than 813,500 citations were added to MEDLINE in 2017. Thus, it becomes an extensive database for biomedical research. The MEDLINE database belongs to a larger family of more than 40 NLM databases called MEDLARS (Medical Literature Analysis and Retrieval System), which includes specialized databases such as Cancerlit, AIDSLINE, and TOXLINE. In process version of MEDLINE is PreMEDLINE which is updated on a daily basis. It provides citations, along with abstracts, which are available before they are indexed for MEDLINE.
Databases associated with health-related research
| Non-academically affiliated | Academically affiliated (possible) |
|---|---|
| PubMed Ovid Medline | Ovid MEDLINE |
| Cochrane Library Cochrane Library | Cochrane library |
| TRIP: Turning Research into Practice | Web of Science |
| SUMSearch | Scopus |
| EMBASE |
Other sources of the biomedical database include EMBASE and Database of Abstracts of Review of Effectiveness (DARE). Various professional organizations also provide access to secondary evidence, clinical recommendations, parameters of care, position papers, or academy statements related to dental practice include,
- American Dental Association, Center for Evidence-Based Dentistry website (http://ebd.ada.org).
- Centers for Disease Control and Prevention: Guidelines and Recommendations (http://www.cdc.gov/OralHealth/ guide lines.htm).
- American Heart Association (AHA): Prevention of Bacterial Endocarditis, Recommendations by the American Heart Association (http://circ.ahajournals.org/cgi/content /full/96/1/358).
- American Academy of Periodontology (AAP): AAP Clinical and Scientific Papers (http://www.perio.org/resources products/posppr2.html).
- American Academy of Pediatric Dentistry: Definitions, Oral Health Policies and Clinical Guidelines (http://www. aapd.org/policies/).
Two journals that provide high-quality articles on evidence-based research are Evidence-Based Dentistry (http://www.nature.com/ebd/index.html) and the Journal of Evidence-Based Dental Practice (http://www.jebdp.com). Both of these journals publish analyzed systematic reviews or original research articles along with expert commentary at the end and description of clinical application.
Know More…
Cochrane Collaboration:
The Collaboration is named after Professor Archie Cochrane, an epidemiologist. He stressed upon the importance of properly evaluating healthcare interventions, particularly through randomized controlled trials. The aim of making this collaboration was to provide accurate and up-to-date information about healthcare effects which is readily available worldwide. The collaboration encompasses more than 28,000 contributors in more than 100 countries. The primary function of Cochrane collaboration is to disseminate member-produced systematic reviews of healthcare interventions through updates of The Cochrane Library. The Cochrane Oral Health Group is but 1 of 52 groups. There are 10 key principles based on which this collaboration functions 12,
1. Collaboration, by internally and externally fostering good communications, open decision making, and teamwork.
2. Building on the enthusiasm of individuals, by involving and supporting people of different skills and back-grounds.
3. Avoiding duplication, by good management and coordination to maximize economy of effort.
4. Minimizing bias, through a variety of approaches such as scientific rigor, ensuring broad participation, and avoiding conflicts of interest.
5. Keeping up to date, by a commitment to ensure that Cochrane Reviews are maintained through identification and incorporation of new evidence.
6. Striving for relevance, by promoting the assessment of healthcare interventions using outcomes that matter to people making choices in health care.
7. Promoting access, by the wide dissemination of the outputs of the Collaboration, taking advantage of strategic alliances, and by promoting appropriate prices, content and media to meet the needs of users worldwide.
8. Ensuring quality, by being open and responsive to criticism, applying advances in methodology, and developing systems for quality improvement.
9. Continuity, by ensuring that responsibility for reviews, editorial processes, and key functions is maintained and renewed.
10. Enabling wide participation in the work of the Colla-boration by reducing barriers to contributing and by encouraging diversity.
Techniques for effectively searching the literature:
The MeSH Vocabulary:
The National Library of Medicine has developed a special vocabulary to index each reference, referred to as MeSH (Medical Subject Headings). The vocabulary contains main headings or index terms. A particular term represents a single concept in the biomedical literature. New terminologies are continuously added to the vocabulary as the new concepts emerge in the scientific literature. Similarly, outdated terminologies are eliminated. When a new article needs to be indexed, trained indexers from the National Library of Medicine choose the MeSH terms (usually 10 to 12) that best characterize the substance of the article (title, authors, source, institution, abstract and more). The main advantage of using vocabulary is to facilitate the retrieval of information from the electronic database whenever required. Along with this, MeSH includes many “entry terms”, which point ………..Contents available in the book……….Contents available in the book……….Contents available in the book……….Contents available in the book……….
Searching the text words:
The ‘text words’ are words or phrases in the title or abstract of the article. The search can also be performed by using these text words. For example, if we are searching for ‘mucositis’ as a subject term, MEDLINE automatically matches it to the MeSH term ‘stomatitis’. As a result, all the articles which have stomatitis as a major concept in the paper will appear in the results of the search. When using the text words, only the articles that have mucositis in their title will appear. But we may get results from medical literature relating to mucosal disorders from other parts of the body such as intestinal mucosal disorders. So, the text words should be searched along with subject terms.
“Exploding” while performing the search:
Sometimes, while searching for a particular term, the search engine comes out with extensive results. This is called “Exploding”. For example, if we search for term ‘inflammation’ we will get enumerable results relating to inflammation. Most of the results obtained may be unwanted. This problem can be avoided by combing searches and applying limits. For example, searching for ‘gingival inflammation’ will restrict the search results to articles related to gingival inflammation.
The text words can also be combined using the Boolean operators AND, OR and NOT to control searches. For example, when we search for ‘periodontitis’ and ‘non-surgical periodontal therapy’ separately, we get a large number of results. However, when these two terms are combined, ‘periodontitis and non-surgical periodontal therapy’, the number of the articles obtained in the results are restricted to a large extent.
Applying limits:
To reduce the number of irrelevant results, appropriate limits can be applied. Although it depends on the question in hand, 25-30 titles should be sufficient to find the relevant results. Most of the databases allow the application of limits such as human vs. animal research, age groups, year of publication, language, publication types and other parameters.
Methodological Filters:
The methodological filters were first developed by members of the evidence-based medicine group at McMaster 13. With the help of these filters, we can combine the tested MeSH terms and text words related to the study design and methodology. The research articles can be retrieved from four categories: therapy, diagnosis, etiology or prognosis.
Appraising the evidence (Appraising)
Critical appraisal is the process of assessing and interpreting evidence through the systematic consideration of its validity, relevance and results. Once the search results have been obtained, one should have the skill to evaluate the evidence found. First of all, the reader should be able to differentiate between clinical guidelines, systematic reviews, meta-analysis and original studies. Further, he/she should critically evaluate the article to find out, if the methods were conducted rigorously and appropriately. Following is a set of questions that can be applied to evaluate a study,
1. Has the study’s aim been clearly stated?
2. Does the sample accurately reflect the population?
3. Has the sampling method and size been described and justified?
4. Have exclusions been stated?
5. Is the control group easily identified?
6. Is the loss to follow-up detailed?
7. Can the results be replicated?
8. Are there confounding factors?
9. Are the conclusions logical?
10. Can the results be extrapolated to other populations?
Internal validity:
Internal validity refers to how well an experiment is done, especially whether it avoids confounding. It indicates to what extent the results of the research may be biased and is thus a comment on the degree to which alternative explanations for the outcome found are possible. Thus, internal validity measures of the strength of the ………..Contents available in the book……….Contents available in the book……….Contents available in the book……….Contents available in the book……….
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
External validity:
External validity is the validity of generalized (causal) inferences in scientific research, that is, is the extent to which the results of a study can be generalized. It is a very important component of research appraisal because it evaluates the generalized applicability of the research.

Applying the evidence in clinical decision making (Applying)
After the search work is complete and the research articles have been critically evaluated, the next step is to apply the outcome in decision making. It must be remembered here that ………..Contents available in the book……….Contents available in the book……….Contents available in the book……….Contents available in the book……….
The clinical guidelines are the recommendations which are applied to the patient based on clinical judgment of the clinician and patient preference. For individual studies, there are three primary questions that need to be answered when determining whether evidence should be applied in practice. The sub-questions in the table help us to decide whether the available evidence is sufficient to be applied in the practice. The answers to these questions tell us whether the results of the study are trustworthy and what is the expected outcome with the patient under treatment.
Questions to be asked when applying the evidence in clinical practice
| Primary question | Sub Question |
|---|---|
| 1. Are the results valid? | Are the studies well designed and executed? What are the types of studies used? |
| 2. What are the results? | What is the certainty of the effect? What is the magnitude of the effect? |
| 3. Can the results be applied to my patient? | Is the population similar? Is the pro- vider similar? Is the setting similar? |
Evaluating the outcome (Assessing)
The last step in evidence-based clinical practice is to evaluate the applied evidence in a specific clinical situation. It is evaluated, how well the treatment plan worked out and how efficiently the endpoint of the treatment was achieved. In other words, was the intended outcome of the treatment was achieved in that particular patient in a given clinical situation or not? More importantly, was the treatment modality applied on the patient was cost-effective and how much time did the whole process take? It has to be critically evaluated in this step that after taking evidence-based decision regarding the treatment plan, did the overall treatment plan and treatment outcome actually benefited the clinician as well as the patient or not.
Barriers to implementing evidence-based clinical practice
There are certain barriers that limit the implementation of evidence-based practice in routine clinical practice. These barriers may be discussed at the practitioner level, at the patient level, and at the organization level.
Practitioner level:
- The most important factor associated with dental practitioners is difficulty in changing current practice model, resistance and criticism from colleagues, and lack of trust in evidence or research.
- Another factor is related to obsolete knowledge and influence of opinion leaders. Some practitioners firmly believe in certain health professional’s views and do not evaluate their views scientifically on the basis of the evidence available.
- Limitation of time availability and organization of the practice may limit the incorporation of EBD in clinical practice.
- The practitioner is not updating his/her knowledge with continuing dental education programs which aim at promoting the quality of life of the patients.
Patient-level:
- Presence of cultural believes regarding the demand for particular treatment by the patients.
- Lack of appropriate education regarding health-related benefits to the patient following implementation of EBD in routine practice.
Organization level:
- At the organizational level, failure to conduct appropriate continuing dental education programs regarding the incorporation of EBD in clinical practice.
- Due to lack of financial resources and defined practice populations.
- Ineffective or unproved activities promoted by health policies.
- At the organizational level, a failure to provide practitioners with access to appropriate information.
Advantages of evidence-based practice
The evidence-based dental practice is beneficial both to the practitioner as well as the patient.
Advantages to the practitioner:
- Improvement in clinical decision making.
- Improved accuracy and greater confidence in treatment planning.
- It ensures maximum treatment safety.
- Provides treatment choices and minimizes risk.
- More effective treatment planning based on the powerful combinations of stronger scientific evidence, clinical judgment and experience, as well as patient preferences and value.
- Improved treatment satisfaction both to the practitioner as well as the patient.
- Enhanced recognition in the community and with peers as a thought leader practice.
Advantages to the patient:
- Improvement in the confidence of the patient in doctor as a result of improved results of the treatments provided.
- Greater incentive to invest in quality oral health care.
Conclusion
There is immense research going on in the field of Periodontology. Thousands of research articles are published every year in various journals. The clinician must be able to identify how to apply this clinical research in his/her general practice. To facilitate this goal, evidence-based Periodontology is the need of the hour. With more and more research coming into the picture every day, it becomes essential to incorporate it into our decision making and treatment planning. Evidence-based decision making incorporates the most current and comprehensive scientific evidence along with the patient’s needs and preferences to make individualized healthcare decisions. It not only helps the clinicians to be confident about the treatment they are rendering to their patients but also helps the patients who are ultimately benefited from the treatment. With an understanding of how to effectively use EBDM, practitioners can quickly and conveniently stay current with scientific findings on topics that are important to them and their patients.
References
References are available in the hard-copy of the book.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

