Introduction to Chlorhexidine (CHX)
Presently, CHX is the most effective anti-plaque and anti-gingivitis agent. It is effective against Gram +ve and Gram -ve bacteria as well as against facultative aerobes and anaerobes 126-128. It has also been shown to inhibit the growth of yeast and fungi 129. Although CHX is not considered virucidal, but it shows some activity on the lipid envelopes of viruses such as HIV, herpes 1 and 2 and influenza A 130. Structurally, it is a cationic poly-biguanide (bisbiguanide) and is used primarily as its salts, dihydrochloride, diacetate, and digluconate because the base molecule is relatively insoluble in water.
Historical aspect
CHX has been used for more than 60 years as an effective plaque control and anti-gingivitis agent. This chemical compound was first discovered by the Imperial Chemical Industries, Limited (Manchester, UK) in 1950 while researching the synthesis of anti-malarial agents 131. Davis et al. (1954) 126 found that a bisbiguanide with a chemical structure 1,6 bis-4 chloro, phenyldiguanidohexane had the greatest bacteriostatic and bactericidal properties. In 1954, they published an article, entitled “1:6-Di-4′-Chlorophenyl-diguanidohexane” (Hibitane): Laboratory Investigation of a New Antibacterial Agent of High Potency. In the same year, CHX gluconate was registered by Imperial Chemical Industries Co. Ltd of Macclesfield (United Kingdom) as Hibitane®, the first internationally accepted antiseptic for cleansing wounds and the skin. In 1957, Chlorhexidine digluconate was first introduced commercially in the United Kingdom as a disinfectant and topical antiseptic.
In the United States, it was first introduced commercially in 1970’s as a disinfectant and topical antiseptic under the name “Hibiclens”, which is 4% CHX gluconate solution primarily used as an antimicrobial skin cleanser. Löe and Schiött (1970) 132 were first to demonstrate the anti-plaque property of CHX, when they introduced 0.2% CHX gluconate mouth rinse and in an experimental gingivitis study, demonstrated that twice or even once oral rinses with 10 ml of 0.2% CHX rinses for 60 seconds prevented plaque growth and development of gingivitis over a period of 21 days in which no oral hygiene measure were used. The first study, which evaluated the application CHX mouth rinse with tooth brushing was carried out by Flotra et al. 133 in 1972, who on a group of soldiers used ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……..
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.
India Users:
International Users:
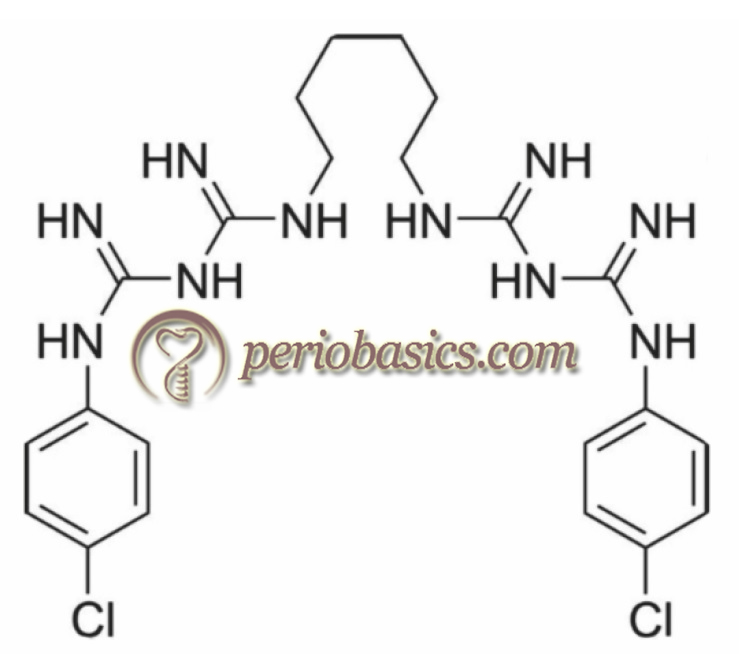
Chemical structure
Structurally, CHX molecule is an amphipathic molecule with both hydrophilic and hydrophobic groups. It is cationic at physiological pH. CHX is made up of two symmetrical chlorophenol rings (4-chlorophenyl) and two biguanide groups, united by a central hydrophobic hexamethylene chain. As already stated, CHX is used in salt form because of the relative insolubility of the base molecule. In the majority of mouth rinses, gels and varnishes digluconate salt is used because it is most water and alcohol soluble. Because of the highly cationic nature of CHX, it is difficult to formulate in dentifrices due to the risk of inactivation of the CHX molecule with other anionic ingredients.
Mechanism of action of CHX
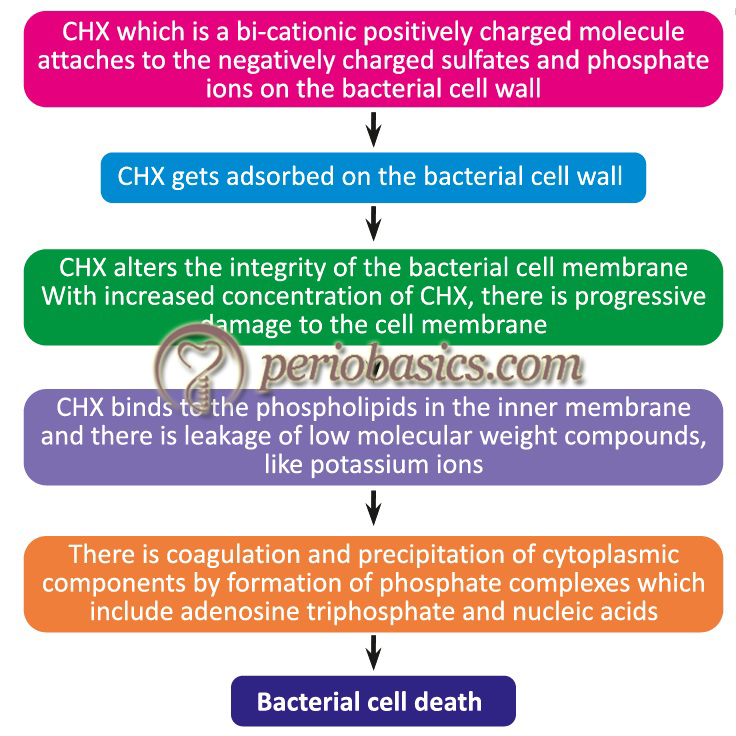
CHX has a broader antibacterial spectrum as well as quicker killing rate than many other antimicrobials 134. CHX is a membrane-active agent. The primary action of CHX is bacterial membrane disruption, causing concentration-dependent growth inhibition and cell death 135. The outermost layer of the bacterial cell wall is negatively charged which is usually stabilized by the presence of cations such as Mg2+ and Ca2+. Because of the negatively charged bacterial surface, most of the cationic antiseptics have a high affinity towards bacterial cell wall 136, 137. The first step in its bactericidal activity is its adsorption onto the wall of the microorganism, which is a very rapid process because of its bi-cationic nature 138. Neutral or slightly alkaline pH favors the CHX adsorption. CHX adsorption also depends on ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……..
After binding to the phospholipids in the inner cell membrane, CHX causes increased membrane permeability altering the metabolic osmoregulatory and the enzyme content of the bacterial cell. The alteration in membrane permeability is responsible for the bacteriostatic activity of CHX. At higher CHX concentrations, there is crystallization of the bacterial cell membrane due to stronger interactions of CHX molecules with the cell membrane. As a result, there is progressively greater damage of membrane, resulting in loss of larger molecular weight compounds and coagulation and precipitation of cytoplasm 140-142. The bacteriostatic and bactericidal concentrations of CHX vary for different bacterial species. CHX is more effective against Gram +ve bacteria (≥ 1 µg/l) as compared to Gram -ve bacteria (10 to >73 μg/ml) due to the difference in their cell wall structure 143.
Mechanisms of plaque inhibition by CHX
There may be multiple mechanisms of plaque inhibition by CHX. Most of the authors 144, 145 have proposed three mechanisms of plaque inhibition by CHX,
1. The blocking of salivary glycoproteins by CHX prevents their binding to the tooth surface, hence inhibiting the acquired pellicle formation.
2. Another proposed mechanism of CHX mediated plaque inhibition is that it reduces the ability of bacteria to bind to the tooth surfaces because of adsorption of CHX to the extracellular polysaccharides in the bacterial cell wall or glycocalyx. This mechanism is supported by findings that the addition of sucrose to bacterial suspensions in vitro reduces the anti-bacterial effect of CHX 144. When sucrose is added to the bacterial suspension, the production of extracellular polysaccharides by bacteria is increased. Hence, a greater amount of the drug is attached to the outer cell coating and less amount of drug acts on the cell membrane, which is required for the bacteriostatic and bactericidal activity of CHX 146.
3. The third proposed mechanism of CHX is that it may ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……..
Pharmacokinetics and Pharmacodynamics of CHX in the oral cavity
The local application of CHX in the form of mouth rinses or irrigation follows principles of first order kinetics, where high initial concentrations of the agent along with multiple applications are required to provide sustained effectiveness. To improve the sustained release of CHX, controlled delivery systems have been designed. Controlled drug delivery system was proposed by Judah Folkman in mid-1960’s. In this system, the drug is released in a controlled manner when a polymer, whether natural or synthetic, is judiciously combined with a drug or other active agent in such a way that the active agent is released from the material in a pre-designed manner 154. The basic components of a controlled delivery system are drug reservoir, the rate-controlling element, and biological platform. These systems are the basis of local drug delivery. Periochip® is CHX chip placed in the periodontal pockets for sustained release of the active agent (discussed in detail in “Local drug delivery in periodontics”).
Since most of the surfaces in the oral cavity are negatively charged; CHX gets well dispersed in the oral cavity. Also, due to its high affinity towards negatively charged surfaces, it is not easily displaced and stays for a longer duration of time which accounts for its good substantivity 155. The attached CHX molecules are slowly displaced by the calcium ions in saliva. However, its high affinity towards negatively charged surfaces is not only responsible for its efficacy, but also contributes to its side effects 156.
The substantivity of CHX depends on its concentration used, rinsing time, temperature and pH 98, 157. 0.2% and 0.12% concentrations of CHX are widely used in mouth rinses. CHX does not penetrate the oral mucosa and most of it is retained on the oral surfaces primarily bound to glycoproteins. The interactions between surface glycoproteins and ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……..
CHX has a good substantivity in the oral cavity and most of the studies have shown its substantivity up to 7 hours following single oral application. To determine substantivity of CHX, plate culture techniques are most commonly used. Studies 160, 161 have reported the immediate antibacterial effect of 0.12% CHX mouth rinse (15 mL solution for 30 seconds) for a minimum of 7 hours. However, the immediate decrease in bacterial viability was 72%-87% (<1 log10) and 58%-88% (<1 log10) at 7 hours after the mouth rinse. Studies done on 0.2% CHX mouth rinse (10 mL/1 minute) have reported the immediate antibacterial effect of CHX with a reduction of ≥90% (≥1 log10) in the bacterial count and a substantivity of up to 7 hours 124, 162-165. Many other studies have also confirmed the immediate antibacterial effect of a single application of 0.12% or 0.2% CHX for up to 7 hours 163, 165-167. However, some authors have reported that there is a continuous fall in the bacterial viability for up to 3-5 hours after the mouth rinse followed by recovery of salivary flora 7 hours after the mouth rinse 168, 169. One study has suggested that CHX persists in the oral cavity for up to 12 hours, which was confirmed by suppressed bacterial count 170.
Know More…
0.12% and 0.2% preparations of CHX:
One of the primary disadvantages of CHX is staining of teeth and soft tissue because of its affinity for dietary compounds, some of which cause staining 149. Sometimes, this staining is so severe that professional prophylaxis is required to remove these stains. Also, tongue brushing may be required to remove the stains from the surface of the tongue 150. Another major side effect of CHX is the altered taste sensation. To overcome these side effects of CHX, its lower concentrations have been formulated such as Peridex (0.12% CHX) and Eludril (0.1% CHX). In one experimental study staining of acrylic specimens were evaluated following the application of 0.2% and 0.1% CHX preparations. The results of the study demonstrated heavy staining with 0.2% CHX whereas 0.1% CHX produced no staining of acrylic specimens greater than that of controls. The anti-microbial effects of both the preparations were similar. The overall activity of CHX in 0.1% CHX preparation was partially or completely inactivated, which, according to the authors was because of the presence of detergent. The authors further stated that the anti-microbial activity of the solution (0.1% CHX) was derived more from the detergent than CHX 151. These findings were supported by another short-term single-blind crossover study in which 0.2% (Corsodyl) and 0.1% (Eludril) preparations of CHX were used on a group of 14 volunteers. During the course of the study, observations were made for plaque formation, gingivitis, and staining. The authors reported significantly less gingivitis in 0.2% CHX group at 12 and 19 days as compared to 0.1% CHX group, but staining was markedly reduced in 0.1% CHX group. The authors concluded that the reduced anti-plaque activity in lower CHX concentration preparation was due to some components in its preparation which inactivated CHX 152.
Some other authors have preferred 0.1% CHX preparation over 0.2% CHX preparation as a prophylactic mouth rinse after oral surgery because of its better patient acceptance and fewer side effects 153.
Synergistic effect of CHX with other agents
CHX has been shown to have synergistic effects when combined with some other agents. The addition of fluoride or thymol in CHX containing varnish has been shown to increase the anti-caries activity of varnish 171. The combination of CHX and copper (Cu2+) has been shown to be synergistic in inhibition of growth of Streptococcus mutans, Actinomyces viscosus, and Actinomyces naeslundii 172. Low concentrations of CHX combined with copper (Cu2+) can be used in a rinse form to reduce caries risk as well the development of gingivitis. Another synergism reported with CHX is with zinc ions. One study reported that Porphyromonas gingivalis proteinases (gingipains) were effectively inhibited by CHX and the inhibitory effects of CHX were enhanced 3 to 30-fold by Zn ions 173. CHX has also been shown to act synergistically with ozone in root canal disinfection. The study reported that application of 2% CHX followed by application of ozone gas for 24 seconds promoted the complete elimination of Candida albicans and Enterococcus faecalis 174. Another study reported the synergistic action of CHX and hydrogen peroxide against Enterococcus faecalis when used as an intracanal irrigant 175.
CHX delivery systems
The CHX formulations are available in various formulations such as mouth rinses, gels, sprays, toothpastes and chewing gums.
Mouthrinses:
These are the most commonly used formulations of CHX. Most of the commercially available formulations contain CHX gluconate solution in a suitable flavored and colored vehicle. The mouth rinses are available in 0.12% or 0.2% CHX concentrations. CHX is bitter in taste and various agents are added in the mouth rinses to make its taste acceptable to the patients. However, masking of the taste by addition of various agents is also associated with the decreased activity of CHX.
Gels:
The CHX preparations in gel form are effective formulations for plaque control, especially in handicapped individuals 176. These can be delivered with the help of a toothbrush or tray. CHX gels are available in both 0.12% and 0.2% preparations. 2% gel formulation of CHX has also been shown to be an effective root canal lubricant. However, the gel formulation required significantly more time for killing microorganisms as compared to the corresponding concentration in liquid form 176.
Sprays:
Both 0.1% and 0.2% CHX sprays are available commercially. These are very useful in physically and mentally handicapped patients. It has been shown that twice-daily spray of non-aerosol CHX delivers approximately 1.4 to 2 ml of CHX which is one-seventh of the optimal mouth rinse dose of 0.2% CHX. However, the effects on plaque and gingival bleeding observed in both cases were the same 28.
Toothpastes:
As already stated, CHX formulation in toothpastes is associated with the problem of CHX inactivation due to its highly cationic nature because of which it can bind to the components in the toothpaste. CHX is inactivated by sodium lauryl sulfate (SLS) which is commonly used as a surfactant in toothpastes. However, toothpastes with high active CHX availability have been formulated and tested for their anti-plaque, anti-gingivitis effectiveness 177. An example of CHX containing toothpaste is Cariax® which has 0.12% CHX.
Chewing gums:
Chewing gums are an effective method for delivering various topical dental prophylactic and therapeutic agents. Chewing gums containing various therapeutic agents such as fluorides, xylitol, metal salts, enzymes and CHX diacetate have been made commercially available by various companies. Most of the CHX containing chewing gums have been formulated in such a way that they deliver unbound active CHX molecules. These are particularly indicated in patients with xerostomia and malodor. It may also be advised as an ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……..
Varnishes:
Varnishes are an effective way of professionally delivering therapeutic agents. It does not require patient’s compliance and the effects can be monitored by the operator. They do not cause discoloration and staining. Presently, commercially available CHX varnishes are EC40, Chlorzoin and Cervitec 178. The advantage of varnish application is that it can provide sustained delivery of CHX, however, it requires repeated application of the varnish 178. Further, it provides caries prevention benefits similar to that of topical fluoride applications in the permanent teeth of teenagers 179. These are also beneficial in preventing root surface caries 180. The application of subgingival CHX varnish in conjunction with root planing has been shown to reduce pocket depth at initially deep sites 181.
Side effects of CHX
Various side effects associated with the use of CHX are, brown discoloration of the teeth, restorative materials and dorsum of the tongue, taste perturbation, increased calculus formation, sometimes there can be dose-dependent oral mucosal erosion which is an idiosyncratic reaction and parotid swelling 2, 182, 183.
CHX staining:
The long term use of CHX is associated with staining. The CHX stains are yellowish-brown and can be seen on the teeth, dorsum of the tongue, composite restorations and on artificial teeth. The possible mechanisms involved in CHX associated staining include non-enzymatic browning (Maillard reaction), the formation of pigmented metal sulfides and precipitation of dietary chromogens 184, 185. The Maillard reaction occurs when amine-containing compounds and carbohydrates react to form colored compounds. Berk suggested that carbohydrates and proteins present in the acquired pellicle may undergo a series of condensation and polymerization reactions which ultimately causes discoloration of the acquired pellicle 186. In the presence of CHX, the formation of acquired pellicle is accelerated and CHX also catalyzes steps in the Maillard reaction 187. This proposed mechanism is further supported by the demonstration of furfurals, intermediate products in Maillard reactions in brown-discolored pellicle 188. However, according to Eriksen et al. (1985) 185, Maillard’s reaction in pellicle accounting for tooth staining is still inconclusive. This staining mechanism has not been agreed upon by many authors for many other antiseptics.
Another proposed mechanism of CHX associated staining is the formation of the pigmented sulfides of iron and tin. It has been suggested that CHX denatures the pellicle because of which the sulfur radicals are exposed. These exposed sulfur radicals react with metal ions to form the metal sulfides responsible of discoloration. Iron and tin ions have been shown to be particularly associated with the formation of metal sulfides. In a study, Warner et al. (1993) 189 showed that levels of iron and sulfur were increased in subjects treated with CHX as compared to water controls. The authors concluded that the chromophore was not a sulfide, but sulfur-containing organic compound and metal ion complex. The authors also stated that the deposition of sulfate proteins was accelerated by CHX.
Many clinical studies have suggested that staining is caused by ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……..
Taste disturbance (dysgeusia):
The aqueous solution of CHX is very bitter which causes altered taste perception. The major taste perceptions being affected are sweet and salt 198. The bi-cationic nature of the CHX molecule which is responsible for its antimicrobial activity seems to be associated with taste disturbance. This taste disturbance is thought to be caused by the denaturation of surface proteins on the taste buds by CHX molecules. The altered taste sensation is transient and it returns to normal after the cessation of the drug.
Oral mucosal erosion, which is an idiosyncratic reaction:
Occasionally, burning sensation and painful desquamative lesions on oral mucosa have been reported. The reason for these oral lesions seems to be precipitation of the mucin layer with a resultant reduction in its lubricating effect 199. These reactions are dose-dependent and resolve after the cessation of the drug.
Increased calculus formation:
The exact mechanism of increased calculus formation with the use of CHX is not well established. However, the local change in pH resulting from adsorption of the cationic agent per se and marked impact on bacterial integrity seems to be the possible causes. The CHX affected bacteria cannot metabolize sugars and thus acids are not produced. Also, in vitro study done by Wasserman et al. (1958) 200 has demonstrated that dead bacteria calcify more rapidly than live microorganisms.
Unilateral or bilateral parotid swelling:
This is a rare side effect of CHX where unilateral or bilateral parotid swelling is observed. The exact mechanism of the precipitation of this condition has not been explained well. However, the involvement of viral infection (parotitis) in connection with CHX mouth rinses has been suggested by some authors that might be co-incidental, but cannot be completely disregarded 133. Another explanation is the inhibition of antiviral activity of secretory IgA. The secretory IgA accumulate on the mucous membrane and probably the precipitation of acidic proteins in the mucin layer which coats the mucous membrane may interfere with the anti-viral activity of IgA 201.
Another proposed cause of CHX associated parotid gland swelling is ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……… Contents available in the book ……..
Uses of CHX other than mouth rinses:
CHX is a very effective anti-microbial agent. It has many uses in dentistry other than mouth rinses. These are,
- It is used for disinfecting hands, skin and washing the wounds.
- It is used in chemical sterilization.
- As an endodontic irrigant. 0.2 % CHX can be used as an endodontic irrigant and intracanal dressing.
- It has been used to reduce the severity of gingival overgrowth.
- Application of bioadhesive 0.2% CHX gel in the socket after 3rd molar extractions can help in reducing the chances of alveolitis (dry socket).
- Treatment of dental unit water lines with 0.12% CHX gluconate reduces bacterial load in dental unit waterlines.
- Pre-treatment rinses with 0.2 or 0.12% CHX are effective in reducing bacterial aerosols.
Conclusion
Chlorhexidine has been considered as the “Gold standard” for mouth rinses. It has many properties that make it a near-ideal mouthwash. Thus, it is the most extensively used mouthwash at present. However, it should be remembered that CHX should not be recommended to each and every patient. As discussed in “Chemical plaque control agents”, there are many chemical agents that can be used to prevent plaque formation in the oral cavity. Hence, the chemical plaque control agents should be prescribed cautiously to the patients.
References
References are available in the hard-copy of the website.
Periobasics: A Textbook of Periodontics and Implantology
The book is usually delivered within one week anywhere in India and within three weeks anywhere throughout the world.

